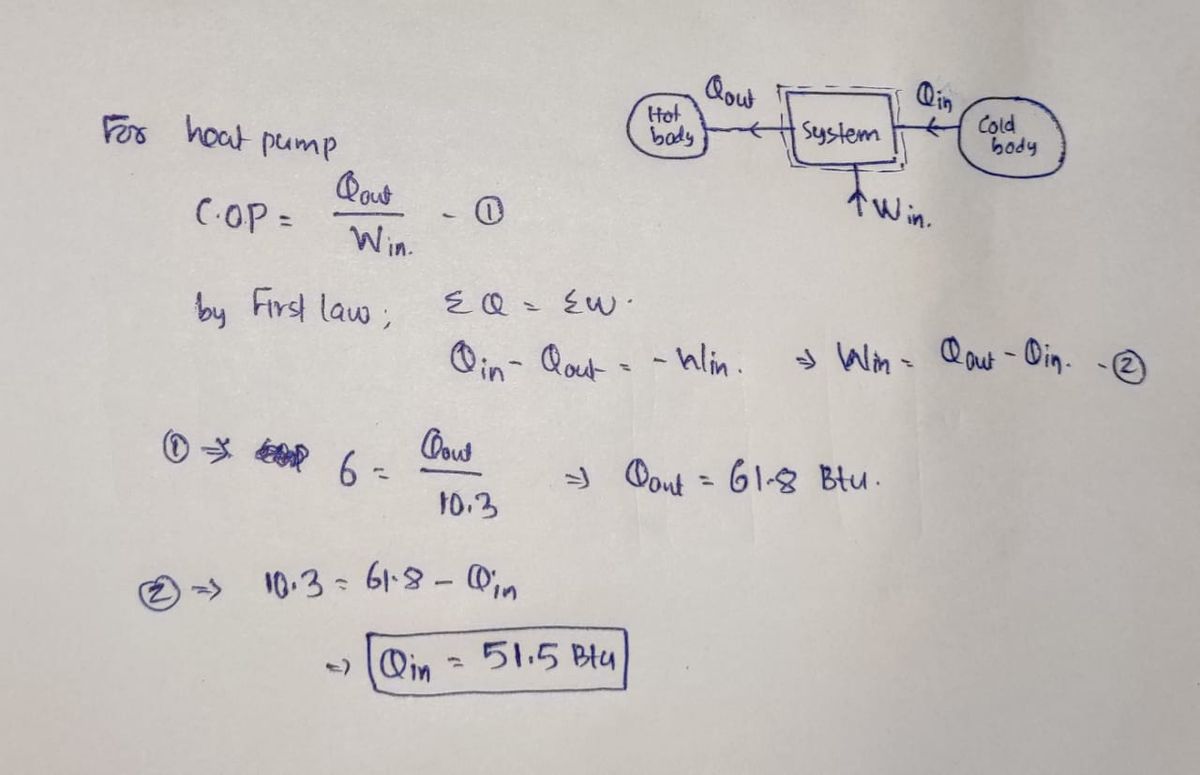
Consider the heat pump cycle outlined above. If the C.O.P. of the cycle is reported to be Y = 6.0 and the work input to the system is found to be W;in = 10.30 Btu, determine the heat transferred into the system during cycle, Qin (Btu). %3D !!
Consider the heat pump cycle outlined above. If the C.O.P. of the cycle is reported to be Y = 6.0 and the work input to the system is found to be W;in = 10.30 Btu, determine the heat transferred into the system during cycle, Qin (Btu). %3D !!
Elements Of Electromagnetics
7th Edition
ISBN:9780190698614
Author:Sadiku, Matthew N. O.
Publisher:Sadiku, Matthew N. O.
ChapterMA: Math Assessment
Section: Chapter Questions
Problem 1.1MA
Related questions
Concept explainers
Question

Transcribed Image Text:Show incorrect Only
Ford: Attempt 5
Qin
Qout
System
Hot Rody
1.
Win
Consider the heat pump cycle outlined above. If the C.O.P. of the cycle is reported to
be Y = 6.0 and the work input to the system is found to be Win = 10.30 Btu,
determine the heat transferred into the system during cycle, Qin (Btu).
!!
%3D
Your Answer:
Answer
units
37°F Clear
bp
17
f8
f9
ho
16
10
14
fs
%24
&
4.
8
近
Expert Solution
Step 1
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Elements Of Electromagnetics
Mechanical Engineering
ISBN:
9780190698614
Author:
Sadiku, Matthew N. O.
Publisher:
Oxford University Press

Mechanics of Materials (10th Edition)
Mechanical Engineering
ISBN:
9780134319650
Author:
Russell C. Hibbeler
Publisher:
PEARSON

Thermodynamics: An Engineering Approach
Mechanical Engineering
ISBN:
9781259822674
Author:
Yunus A. Cengel Dr., Michael A. Boles
Publisher:
McGraw-Hill Education

Control Systems Engineering
Mechanical Engineering
ISBN:
9781118170519
Author:
Norman S. Nise
Publisher:
WILEY

Mechanics of Materials (MindTap Course List)
Mechanical Engineering
ISBN:
9781337093347
Author:
Barry J. Goodno, James M. Gere
Publisher:
Cengage Learning

Engineering Mechanics: Statics
Mechanical Engineering
ISBN:
9781118807330
Author:
James L. Meriam, L. G. Kraige, J. N. Bolton
Publisher:
WILEY