2. The following temperature/composition data were obtained for a mixture of Octane (0) and methylbenzene (M) at 1atm. T (°C) 110.9 112.0 114.0 115.8 117.3 119.0 121.1 123.0 | Xm (1) Хм (в) The boiling points are 110.6°C and 125.6°C for M and O respectively. (a) Plot the temperature/composition diagram for the mixture. What is the composition of the vapour in 0.908 |0.795 0.615 0.527 0.408 0.3 0.203 0.097 0.923 0.836 0.698 0.624 0.527 0.410 0.297 0.164 equilibrium with the liquid of composition (b) xm(1)= 0.250 and (c) xo (1)= 0.250?
2. The following temperature/composition data were obtained for a mixture of Octane (0) and methylbenzene (M) at 1atm. T (°C) 110.9 112.0 114.0 115.8 117.3 119.0 121.1 123.0 | Xm (1) Хм (в) The boiling points are 110.6°C and 125.6°C for M and O respectively. (a) Plot the temperature/composition diagram for the mixture. What is the composition of the vapour in 0.908 |0.795 0.615 0.527 0.408 0.3 0.203 0.097 0.923 0.836 0.698 0.624 0.527 0.410 0.297 0.164 equilibrium with the liquid of composition (b) xm(1)= 0.250 and (c) xo (1)= 0.250?
Introduction to Chemical Engineering Thermodynamics
8th Edition
ISBN:9781259696527
Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Chapter1: Introduction
Section: Chapter Questions
Problem 1.1P
Related questions
Question

Transcribed Image Text:2. The following temperature/composition data were obtained for a mixture of Octane (0) and
methylbenzene (M) at 1atm.
T (°C)
110.9
112.0
114.0
115.8
117.3
119.0
121.1
123.0
| Xm (1)
Хм (в)
The boiling points are 110.6°C and 125.6°C for M and O respectively. (a) Plot the
temperature/composition diagram for the mixture. What is the composition of the vapour in
0.908
|0.795
0.615
0.527
0.408
0.3
0.203
0.097
0.923
0.836
0.698
0.624
0.527
0.410
0.297
0.164
equilibrium with the liquid of composition (b) xm(1)= 0.250 and (c) xo (1)= 0.250?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 7 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
What does the gap means between the two lines of the graph (which is represented by the yellow marker)? Please explain briefly.
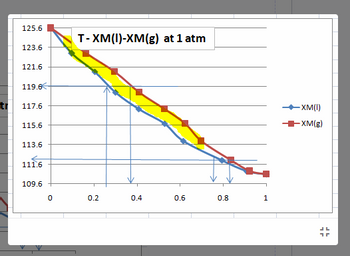
Transcribed Image Text:tr
125.6
123.6
121.6
119.6
117.6
115.6
113.6
111.6
109.6
0
T-XM(I)-XM(g) at 1 atm
0.2
0.4
0.6
0.8
1
-XM(I)
-XM(g)
=
Solution
Follow-up Question
What happens to the trend of the boiling point of the mixture when the amount of octane increase.
Solution
Follow-up Question
Cite some important discussion analysis based on the graph and data which involves the concept of distillation
Solution
Follow-up Question
What does the concept behind the graph wherein the two points of the line meets at the two end. Please explain briefly
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall

Introduction to Chemical Engineering Thermodynami…
Chemical Engineering
ISBN:
9781259696527
Author:
J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark Swihart
Publisher:
McGraw-Hill Education

Elementary Principles of Chemical Processes, Bind…
Chemical Engineering
ISBN:
9781118431221
Author:
Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:
WILEY

Elements of Chemical Reaction Engineering (5th Ed…
Chemical Engineering
ISBN:
9780133887518
Author:
H. Scott Fogler
Publisher:
Prentice Hall


Industrial Plastics: Theory and Applications
Chemical Engineering
ISBN:
9781285061238
Author:
Lokensgard, Erik
Publisher:
Delmar Cengage Learning

Unit Operations of Chemical Engineering
Chemical Engineering
ISBN:
9780072848236
Author:
Warren McCabe, Julian C. Smith, Peter Harriott
Publisher:
McGraw-Hill Companies, The