
Draw the structure of hexanoic acid. Label the alpha carbon.
Interpretation:
The structure of hexanoic acid is to be drawn. The alpha carbon is to be labeled.
Concept introduction:
Carboxylic acids come under the classification of organic acids. They contain a carboxyl group and a hydroxyl group attached to each other. The general formula for carboxylic acids is
Answer to Problem 19.1E
The structure of hexanoic acid with the labeled alpha carbon is shown below.
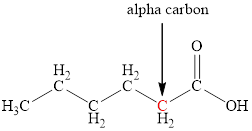
Explanation of Solution
Carboxylic acids are named by adding the suffix –oic acid to the name of the base alkane. The name of hexanoic acid suggests that the base alkane is six carbon atoms long. The alpha carbon is the one which is directly attached to the carbonyl group. Therefore, the structure of hexanoic acid with the labeled alpha carbon is shown below.

Figure 1
The structure of hexanoic acid with the labeled alpha carbon is shown in Figure 1.
Want to see more full solutions like this?
Chapter 19 Solutions
Chemistry for Today: General, Organic, and Biochemistry
- Amino acids are solids with relatively high decomposition points at normal temperature. Please explain why.arrow_forwardDescribe how you would determine if a saccharide has an alpha or beta configuration?arrow_forwardWhat functional groups are present in a carbohydrate molecule? a. Carboxyl and carbonyl groups b. Alcohol and carboxyl groups c. Hydroxyl and carbonyl groups d. Hydroxyl and hydrogen groupsarrow_forward
- Give the name of the molecule. Circle and label the base and sugar parts of the molecule. Number the carbon atoms of the sugar in the conventional way. Identify all chiral atoms in the molecule. Label a carbonyl oxygen, and a primary and secondary amine group.arrow_forwardFill in the blanks to complete the reaction.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





