
Using Figure 3.1, determine
a. the number of grams of
b. the number of grams of water required to dissolve
c. the number of grams of water required to dissolve
d. the number of grams of water required to dissolve a mixture containing
(a)
Interpretation:
The number of grams of
Concept introduction:
Solution is a homogenous mixture of two or more components. A sample taken from any part of the solution will have the same composition as the rest of the solution. Many chemical reactions occur in water solutions.
Answer to Problem 1ASA
The number of grams of
Explanation of Solution
The given illustration of the graph is shown below.
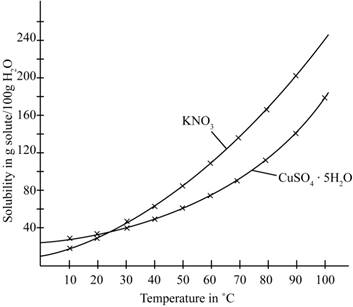
Figure 1
According to the above graph, the number of grams of
The number of grams of
(b)
Interpretation:
The number of grams of water required to dissolve
Concept introduction:
Solution is a homogenous mixture of two or more components. A sample taken from any part of the solution will have the same composition as the rest of the solution. Many chemical reactions occur in water solutions.
Answer to Problem 1ASA
The number of grams of water required to dissolve
Explanation of Solution
The given illustration of the graph is shown below.
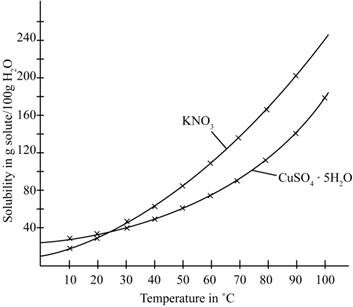
Figure 1
According to the above graph, the number of grams of
Thus, water required to dissolve
The number of grams of water required to dissolve
(c)
Interpretation:
The number of grams of water required to dissolve
Concept introduction:
Solution is a homogenous mixture of two or more components. A sample taken from any part of the solution will have the same composition as the rest of the solution. Many chemical reactions occur in water solutions.
Answer to Problem 1ASA
The number of grams of water required to dissolve
Explanation of Solution
The given illustration of the graph is shown below.
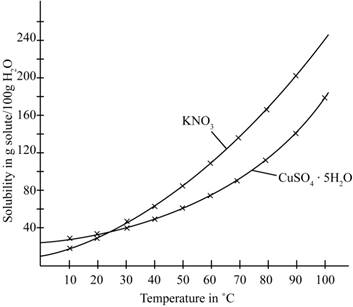
Figure 1
According to the above graph, the number of grams of
Thus, the water required to dissolve
The number of grams of water required to dissolve
(d)
Interpretation:
The number of grams of water required to dissolve a mixture containing
Concept introduction:
Solution is a homogenous mixture of two or more components. A sample taken from any part of the solution will have the same composition as the rest of the solution. Many chemical reactions occur in water solutions.
Answer to Problem 1ASA
The number of grams of water required to dissolve a mixture containing
Explanation of Solution
The given illustration of the graph is shown below.
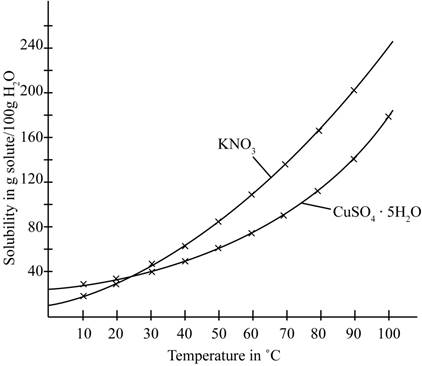
Figure 1
The amount of water required to dissolve
The amount of water required to dissolve
Therefore, the total amount of water required to dissolve a mixture containing
The number of grams of water required to dissolve a mixture containing
Want to see more full solutions like this?
Chapter 3 Solutions
Chemical Principles in the Laboratory
- The dispersed phase of a certain colloidal dispersion consists of spheres of diameter 1.0 102 nm. (a) What are the volume (V=43r2) and surface area (A = r2) of each sphere? (b) How many spheres are required to give a total volume of 1.0 cm3? What is the total surface area of these spheres in square meters?arrow_forwardFor each of the following pairs of solutions, select the solution for which solute solubility is greatest. a. Ammonia gas in water with P = 1 atm and T = 50C Ammonia gas in water with P = 1 atm and T = 90C b. Carbon dioxide gas in water with P = 2 atm and T = 50C Carbon dioxide gas in water with P = 1 atm and T = 50C c. Table salt in water with P = 1 atm and T = 60C Table salt in water with P = 1 atm and T = 50C d. Table sugar in water with P = 2 atm and T = 40C Table sugar in water with P = 1 atm and T = 70Carrow_forward6-16 Answer true or false. (a) Solubility is a physical property like melting point and boiling point. (b) All solutions are transparent—that is, you can see through them. (c) Most solutions can be separated into their components by physical methods such as distillation and chromatography.arrow_forward
- For each of the following pairs of solutions, select the solution for which solute solubility is greatest. a. Oxygen gas in water with P = 1 atm and T = 10C Oxygen gas in water with P = 1 atm and T = 20C b. Nitrogen gas in water with P = 2 atm and T = 50C Nitrogen gas in water with P = 1 atm and T = 70C c. Table salt in water with P = 1 atm and T = 40C Table salt in water with P = 1 atm and T = 70C d. Table sugar in water with P = 3 atm and T = 30C Table sugar in water with P = 1 atm and T = 80Carrow_forward4. A solution is created by dissolving 74g of NaNO3 into enough water to make 3.50L of solution. Calculate the m/v% concentration of NaNo3 in this solution. Use stoichiometry conversion factors and actually put divider lines in between the numbers. The answer should be 2.1% m/V. 5. A 2.00L bottle of mineral water has a concentration of %0.00162 KCl mass/volume. Calculate the mass inmilligrams (mg) of KCl present in the bottle. Use stoichiometry conversion factors and actually put divider lines in between the numbers. The answer should be 32.4mg Please answer 4 and 5.arrow_forwardPart 3. Solubility. For the following molecules label whether you think it will be more soluble in water (H₂O) or dichloromethane (CH₂Cl₂) at 25 °C. OH НО. О ОН ОН OH OHarrow_forward
- Step 1: In tube 1, you add 100 ul of plasma to 900 ul of distilled water. Step 2: In tube 2, you then take 100 ul from tube 1 and add 900 ul of distilled water. Step 3: In tube 3, you then take 100 ul from tube 2 and add 900 ul of distilled water. What is the final/total dilution factor in tube 3 at the end of step 3?arrow_forward3. a. A solution contains 1.75 g of a non-volatile, non-clectrolyte solute dissolved in 23.47g stearic acid. The freezing point of the solution is 67.2°C. The freezing point of pure stearic acid is 69.3°C and its freezing point constant, Kp, is 4.5°C kg/mol. What is the molar mass of the solute? b. When a liquid is in a narrow tube, the surface of the liquid, the meniscus, may be either concave, A, or convex, B What does the shape of the meniscus tell you about the intermolecular forces of attraction between the liquid and the tube?arrow_forwardA certain liquid X has a normal freezing point of 7.10°C and a freezing point depression constant =Kf3.27·°C·kgmol−1. A solution is prepared by dissolving some ammonium chloride (NH4Cl) in 700.g of X. This solution freezes at 6.4°C. Calculate the mass of NH4Cl that was dissolved. Round your answer to 1 significant digit. __garrow_forward
- .Prepare standard Fe solutions from a stock solution for which you will need to: determine the concentration in ppm Fe. The exact mass used of the iron-containing compound will be written on the bottle, so you will need to recalculate this with the correct number in the lab, but if,for example, the bottle is labeled 174.3mg of Fe(NH4)2(SO4)2•6H2O in 1.000 liter, what is the concentration in ppm Fe (Hint: this is a concentration in terms of mass, it is not simply 1ppm whole compound = 1ppm Fe.) Thank you!arrow_forwardShow the complete solution. 1. 3H2 (g) + O3 (g) → 3H2O 2. N2O (g) + NO (g) → 3NO3. CaC2 (s) + 2H2O (l) → Ca(OH)2 (s) + C2H2 (g)arrow_forward100 90 NANO, 80 70 60 CaCl, 50 Pb(NO3)2 40 KCI NaCl 30 20 KCIO, 10 0. Ce,(SO 10 20 30 40 50 60 70 80 90 100 Temperature (°C) How many grams of K2Cr207 will dissolve in 100g of water at 80°C? Your Answer: Answer units Solubility (g of salt in 100 g H,O) KNO3 K,Cro,arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





