
Concept explainers
Figure 4.6 Look at each of the processes shown and decide if it is endergonic or exergonic.

To analyze:
The process shown in the figure is endergonic or exergonic.
Introduction:
The endergonic process is the one which is accompanied by the absorption of energy from the surrounding, while exergonic processes is accompanied by the release of energy into the environment.
Explanation of Solution
Given:
Figure (a) 
Figure (b) 
Figure (c) 
Figure (d) 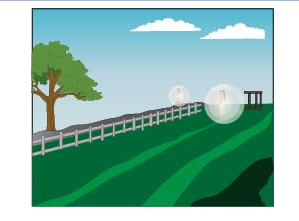
The first figure is of a compost. Decomposition of dead and decaying matter to produce compost or humus is an exergonic process because it is accompanied by the release of heat energy.
The second picture is of a chick coming out from an egg. It is an endergonic process because the process has taken in the energy from the chick.
The third picture is of tea dissolving in water, which is an exergonic process.
The last one is of a ball rolling downwards, in this case, the energy is dissipiated in the environment, which is an exothermic or exergonic process.
Decomposition, dissolving of tea in water and rolling of the ball are exergonic reactions, while the hatching of a chick from an egg is an example of an endergonic reaction.
Want to see more full solutions like this?
Chapter 4 Solutions
Concepts of Biology
Additional Science Textbook Solutions
College Physics
Biology: Life on Earth (11th Edition)
Fundamentals of Anatomy & Physiology Plus Mastering A&P with eText - Access Card Package (10th Edition) (New A&P Titles by Ric Martini and Judi Nath)
Campbell Essential Biology (7th Edition)
- Give the products of each of the following processes. the words on the top will match up with the words on the bottom.arrow_forwardWhich of the following is the best description of the process shown in the image below?arrow_forwardIn the experimental setup described in the attached figure (Figure 2.5 of the textbook), the middle panel shows two compartments with 10 mM KCI on the left (inside) and 1 mM KCL on the right (outside), separated by a membrane that is permeable to K+. What would initially happen if you replaced the KCI solutions with NaCl solutions (1 mM on the left or inside and 10 mM on the right or outside)? (A) Inside 1 mM KC Voltmeter Outside 1 mM KCI Permeable to K Nonet Bux of K (B) Initial conditions Initially Inside Outside 10 mM KC 1 mM KC Net flux of K from inside in outside → At equilibrium --58 mY Inside Outside 10 mM KCI 1 mM KC Flux of K from inside to outside balanced by opposing membrare potential Membrane potential (A) -116 [KL] 4 tended change O Na+ would move up its concentration gradient from the left (inside) to the right (outside) compartment. O Na+ would move down its concentration gradient from the right (outside) to the left (inside) compartment. O Na+ would not move because…arrow_forward
- Why is it reasonable to expect that control can be exerted near the end of a pathway as well as near the beginning?arrow_forwardComplete this mechanismarrow_forwardThe enzyme catalase catalyzes a reaction that breaks down hydrogen peroxide (H₂O2), a toxic chemical that is produced as a by-product of many cellular reactions, into two harm- less substances: water and oxygen. The reaction is shown below. → 2H₂O + O₂ 2H₂O2 This reaction is important to cells because hydrogen peroxide is a by-product of many nor- mal cellular reactions. (a) An experiment can be designed to test the effect of different temperature levels on the functioning of catalase. Identify the appropriate independent and dependent variables for this experiment. (b) Identify an appropriate method to measure the dependent variable in this experiment and justify your choice. Respond in 4 to 6 complete sentences.arrow_forward
- Identify the two cellular process responsible for products described in the figure. Explain the role of each product listed in the processes. refer to picturearrow_forwardPlease help me with part one questions 3,4,5 Thank you PDF link https://www.cusd80.com/cms/lib/AZ01001175/Centricity/Domain/8922/eofad.pdfarrow_forwardWhat is meant by: “The hierarchy of cell sorting?” b) Discuss this in the context of the thermodynamic model of cell interactions (use examples)arrow_forward
- An enzyme is found that catalyzes the reaction X ⇌ Y. Researchers find that the Km for the substrate X is 4 μM, and the kcat is 20 min−1.(a) In an experiment, [X] = 6 mM, and V0 = 480 nM min−1. What was the [Et] used in the experiment?(b) In another experiment, [Et] = 0.5 μM, and the measured V0 = 5 μM min−1. What was the [X] used in the experiment?(c) The compound Z is found to be a very strong competitive inhibitor of the enzyme, with an α of 10. In an experiment with the same [Et] as in (a), but a different [X], an amount of Z is added that reduces V0 to 240 nM min−1. What is the [X] in this experiment?(d) Based on the kinetic parameters given above, has this enzyme evolved to achieve catalytic perfection? Explain your answer briefly, using the kinetic parameter(s) that define catalytic perfection.arrow_forwardGive typed solution onlyarrow_forwardIn the scheme below which represents the mechanism of action for a large number of enzymes: A+B⟺AB⟶C The steady state approximation is reached when: d[AB]/dt≈0 k2≫k1 k−1≫k1 k−1=k1arrow_forward
 Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College
Concepts of BiologyBiologyISBN:9781938168116Author:Samantha Fowler, Rebecca Roush, James WisePublisher:OpenStax College Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning



