Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A:
Q: 1 2 3 4 5 6 7 8 9 89 check on the appropriate box. The first five substances were done for you.…
A: Given different molecules which have intermolecular forces
Q: According to the following reaction, how many grams of hydrochloric acid will be formed upon the…
A:
Q: Which statement comparing neon and sodium is correct? Neon has a larger atomic radii and lower…
A:
Q: in class we learned that some of the groups of the periodic chart have older "common names" as shown…
A: Given that, The given options are B Se C Ca As We have to tell among the given elements which one…
Q: Identify the Grignard reagent(s) that could be used to prepare 5-phenyl-5-nonanol. Select all that…
A: Organic reactions are those in which organic reactant react to form organic products. In the given…
Q: Give detailed Solution with explanation needed (no need Handwritten answer)Give the IUPAC name
A: Rules for IUPAC naming :- Select the longest chain which contains carbon-carbon single bonds.…
Q: How many grams of Ag₂CO3 will precipitate when excess K₂CO3 solution is added to 65.0 mL of 0.746 M…
A:
Q: a. Suppose the reaction system CH4 (g) +202(g) → CO₂(g) + 2H₂O (1) has already reached equilibrium.…
A:
Q: A solution is made by dissolving 0.577 mol of nonelectrolyte solute in 781 g of benzene. Calculate…
A: According to the question, The Moles of non electrolyte solute (benzene) is given by = 0.577 mol…
Q: 18. 19. 2. H3C 3. 1 H H₂CO H CH3 3 -CH₂ H3C- III. CONCEPT QUESTIONS. Select the best answer to the…
A: Since you have asked multiple questions, we will answer the first one for you. To get remaining…
Q: 6. Identify the following reaction types (e.g. condensation, addition, etc.). Y dilute NaOH OH Conc.…
A: we have to determine the type of reaction shown
Q: Consider the following reactions: A=B, K₁=6.02 A C, K₂=2.00 What is K for the reaction C = B?
A:
Q: How long will it take for the concentration of A to decrease from 0.910 M to 0.239 for the reaction…
A:
Q: Consider the reaction data. A products T (K) k (S-¹) 275 0.370 875 0.675 What two points should be…
A: Answer: This question is based on Arrhenius equation which is shown below: lnk=-EaR×1T+lnA
Q: For the reaction A+B+C→D + E, the initial reaction rate was measured for various initial…
A: Given:-
Q: If 0.219 g of a solute is dissolved into 7.832 g of t-butanol (normal freezing point is 25C), and…
A: If 0.219 g of a solute is dissolved into 7.832 g of t-butanol (normal freezing point is 25C), and…
Q: For the reaction system C(s) + H₂O(g) H₂(g) + CO(g) which has already reached state of equilibrium,…
A: Le Chateliar's Principle: The position of equilibrium shifts in a direction that nullifies the…
Q: Write the balanced COMPLETE ionic equation for the reaction when aluminum nitrate and sodium…
A: the chemical reaction can be expressed in a molecular equation or an ionic equation. The molecular…
Q: 3. Which solution is more concentrated? Justify your answer with calculations. OR 00.g of…
A: To determine which solution is more concentrated, we can calculate the molarity of each solution.
Q: OOOO A) HO B) H₂C C) H D) H " "I for it. H₂C Cl HO HO CI CH₂CI OA) D) Which one is the enantiomer of…
A:
Q: Write the balanced NET ionic equation for the reaction when AlCl3 and NaOH are mixed in aqueous…
A: Write the balanced net ionic equation for the given reaction.
Q: What two points should be plotted to graphically determine the activation energy of this reaction?…
A: Activation Energy: the minimum amount of energy that is required to activate atoms or molecules to a…
Q: Write a balanced chemical equation based on the following description: liquid hydrogen peroxide,…
A: Given that, liquid hydrogen peroxide, H2O2 (l), reacts with the trioxygen gas, to produce oxygen gas…
Q: Construct the expression for Kp for the following reaction. 2 C8H18 (1) + 25 O₂(g) = 16 CO₂(g) + 18…
A:
Q: If the equilibrium constant of a given reaction is 2.15, what is the equilibrium constant of its…
A: Characteristics of equilibrium constant are : - If the reaction is reversed, the value of the…
Q: The rate of a certain reaction is given by the following rate law: rate=k[N₂] ² [H₂]² Use this…
A: Rate law of a reaction can be written as- Rate=K[A]x[B]y where, K=rate constant x=order with respect…
Q: 1. Circle the chiral molecules from the following list. CH3 lot I d -CH3 c) e) a a) b) OH I+ H3C f)…
A: Chiral center :- The center or carbon atom in molecule which is connected with four different…
Q: For the following electron-transfer reaction: Co(s) + Cu²+ (aq) Co2+ (aq) + Cu(s) The oxidation…
A: Oxidation is a process which involves loss of electron(s) by an atom or ion, either free or in the…
Q: Try Again Your answer is wrong. Please read the Explanation for more help. Highlight each chiral…
A: Chiral centre Atom especially in an organic molecule that has four different atoms or groups…
Q: In a face-centered cubic lattice, an atom laying in a face of a unit cell is shared equally by how…
A: Since, In fcc, 8 Atoms are present at the corner and 6 atoms present at the face. Thus,
Q: O₂N OH OH Br₂ high temp A reaction does occur! NaOH (1 equiv) Mel (1 equiv) bios oinotluser label E
A:
Q: 3. Given Molecular Equation for reaction of aqueous ammonia with acetic acid solution, based on…
A: Given , Reaction : NH3(aq) +HC2H3O2(aq) → NH4C2H3O2 (aq)
Q: CI C There are no chiral centers. X 3 C
A: This problem is based on general organic chemistry and we have to find out the chiral centre . The…
Q: wouldn't 2d be ~50 C because its the liquid boiling?
A: From the graph , the boiling point of pure liquid A is 50oC and that of pure liquid B is 120oC
Q: (Q11) In the empirical formula experiment, burning Mg in air gives magnesium oxide and magnesium…
A:
Q: Write the acid-base chemical reaction between the two species listed, then label the conjugate…
A:
Q: Consider the following reaction coordinate diagram. 50 45 40 35 IM 30 25 20 15 10 Energy (kJ) 5 0…
A:
Q: 13. 14. 15. CH3 Br CH3 Br O₂N. H== NaHCO3 OH B OH NH CH3 PdCl3(PPH3)2 (cat) IPA/2M Na₂CO3 heat…
A: Given that, the reactions are shown below We have to give the structure of the product. N.B.:…
Q: The Solubility Product Constant for silver hydroxide is 2.0 × 10-8. The molar solubility of silver…
A: We have to calculate the solubility
Q: How do you know when to use which percent enantiomeric excess equation?
A: An enantiomeric mixture known as a racemic mixture has an equal proportion of (R)- and…
Q: At night, almost all NOx is present as NO2. (Ie the NO/NOx concentration is very small). Explain why
A: Air pollution is a significant environmental issue that poses a threat to both human health and the…
Q: Use Cahn-Ingold-Prelog A HO D rules to assign priority to the substituents on this molecule. B H C
A: The Cahn-Ingold-Prelog (CIP) rule is a set of guidelines used to determine the relative priority of…
Q: Using the the Ka of HXO, (Ka 1.2 X 10), determine the identity of the acid if 20 g of HXO, is…
A:
Q: Express Ksp in terms of molar solubility, x, for the salts with the given formulas. AB(s) Ksp = AB₂…
A: This answer provides expressions for the solubility product constant (K_sp) in terms of the molar…
Q: Suppose a reaction with activation energy of 34.0 kJ/mol occurs at 30°C with a rate constant of 1.2…
A:
Q: Complete the balanced molecular chemical equation for the reaction below. If no reaction occurs,…
A: Balanced molecular equation: Balanced molecular equation can be define as a reaction in which number…
Q: A chemist prepares a solution of barium chloride (BaC12) by measuring out 12.9 g of barium chloride…
A:
Q: Calculate the pH of a solution that has pH= as [H₂0*]=6.4 × 10² N M. Round your answer to 2 decimal…
A: Given data : [H3O+] = 6.4 * 10-2 M Now , we use following formula to calculate pH of a given…
Q: At a certain temperature the rate of this reaction is first order in NH OH with a rate constant of…
A:

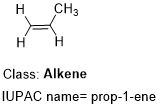
An alkene is characterised by presence of a double bond.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


