Determine if the following reaction will occur at room temperature (298 K) by calculating \Delta G: (hint - look - up Hf, and change its sign to 'reverse' the reaction) H2O(1) H2(g) + 1/2O2(g)
Determine if the following reaction will occur at room temperature (298 K) by calculating \Delta G: (hint - look - up Hf, and change its sign to 'reverse' the reaction) H2O(1) H2(g) + 1/2O2(g)
Oh no! Our experts couldn't answer your question.
Don't worry! We won't leave you hanging. Plus, we're giving you back one question for the inconvenience.
Submit your question and receive a step-by-step explanation from our experts in as fast as 30 minutes.
You have no more questions left.
Message from our expert:
Our experts need more information to provide you with a solution. The enthalpy of the reaction is not given. Please resubmit your question, making sure it's detailed and complete. We've credited a question to your account.
Your Question:
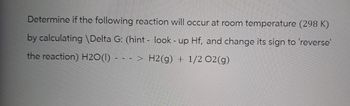
Transcribed Image Text:Determine if the following reaction will occur at room temperature (298 K)
by calculating \Delta G: (hint - look - up Hf, and change its sign to 'reverse'
the reaction) H2O(1)
H2(g) + 1/2O2(g)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning
