
Biochemistry
6th Edition
ISBN: 9781305577206
Author: Reginald H. Garrett, Charles M. Grisham
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
From your Lineweaver-Burk plot, derive the values for the following table. Indicate the appropriate units. (b) By describing the potential changes in the kinetic parameters, identify and justify the type of inhibitor that was inhibiting the acid phosphatase activity.
|
|
Km |
Vmax |
|
Uninhibited |
|
|
|
Inhibited |
|
|
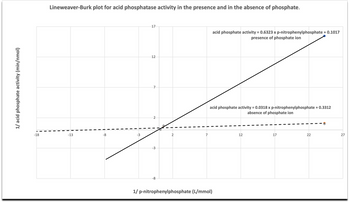
Transcribed Image Text:1/ acid phosphate activity (min/nmol)
-18
Lineweaver-Burk plot for acid phosphatase activity in the presence and in the absence of phosphate.
-13
-8
-3
17
12
7
2
-3
-8
2
7
acid phosphate activity = 0.6323 x p-nitrophenylphosphate + 0.1017
presence of phosphate ion
acid phosphate activity = 0.0318 x p-nitrophenylphosphate + 0.3312
absence of phosphate ion
1/ p-nitrophenylphosphate (L/mmol)
I
12
17
22
27
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 3 steps

Knowledge Booster
Similar questions
- 13. 0.9% (m/v) NaCI solution and 5% (m/v) glucose solution are both isotonic to red blood cells. SHOW your work and watch sig figs & units. c. convert the concentration from M to % (m/v) for a 0.342 M NaC solution. (HINT: convert to g/ml and then multiple by 100%)arrow_forwarduizzes/67365/take Based on the image below, select the correct statements. Note: There may be more than 1 correct response. I Ribose 5-phosphate ribose phosphate pyrophosphokinase (PRPP synthetase) glutamine-PRPP amidotransferase adenylosuccinate synthetase AMP > 5-Phosphoribosylamine I adenylosuccinate PRPP lyase 9 steps Adenylosuccinate AMP IMP <-- ADP - AMP <-- GMP <-- IMP IMP dehydrogenase <- GMP - XMP ADP ATP GMP يمد XMP-glutamine amidotransferase Increased levels of ADP inhibit the production of PRPP. Increased levels of GMP inhibit the production of XMP. O Increased ADP activates PRPP synthase to increase PRPP levels. Increased IMP activates glutamine-PRPP amidotransferase to further increase IMP levels. 8 OBCarrow_forwardThe equilibrium constant for the hydrolysis of the peptide alanylglycine (Gly-Ala in the reaction from Part B) by a peptidase is K = 9.0 × 10² at 310 K. Calculate AG for this reaction. Express the Gibbs free energy to three significant figures. AG = Submit ΠΑΠΙ ΑΣΦ Request Answer ? kJ/mol Keq [Gly] [Ala] [Gly-Ala]arrow_forward
- Intramitochondrial ATP concentrations are about 5 mM, and phosphate concentration is about 10 mM. If ADP is five times more abundant than AMP, calculate the molar concentrations of ADP and AMP at an energy charge of 0.85. Calculate AG for ATP hydrolysis at 37 °C under these condi- tions. The energy charge is the concentration of ATP plus half the concen- tration of ADP divided by the total adenine nucleotide concentration: [ATP] + 1/2[ADP] [ATP] + [ADP] + [AMP]arrow_forwardThe hexapeptide Ala-Met-Leu-Lys-Phe-Asp is digested in the same tube with both Cyanogen Bromide and Trypsin at the same time at pH=7. Draw the structure of the product(s) that would bind to a cation exchange column. (Relevant pKa values are 2.2, 3.9, 9.5 and 10.5. Assume pKa values for any newly generated a-amino and a-carboxyl groups are 9.5 and 2.2 respectively)arrow_forwardCalculate the actual, physiological AG for the reaction Phosphocreatine + ADP = creatine + ATP at 37 °C, as it occurs in the cytosol of neurons, where [phosphocreatine] = 4.7 mM, [creatine] = 1.0 mM, [ADP] = 0.73 mM, and [ATP] = 2.6 mM. Standard Free Energies of Hydrolysis of Some Phosphorylated Compounds Phosphorylated compound AGʻ° (kJ/mol) phosphoenolpyruvate -61.9 phosphocreatine -43.0 ADP (→ AMP + P;) -32.8 ATP (→ ADP + P;) -30.5arrow_forward
- The ΔG°′ for hydrolytically removing a phosphoryl group from ATP is about twice as large as the ΔG°′ for hydrolytically removing a phosphoryl group from AMP (−14 kJ · mol−1). Explain the discrepancy.arrow_forwardHuman blood serum contains a class of enzymes known as acid phosphatases, which hydrolyze biological phosphate esters under slightly acidic conditions (pH 5.0): R-O-P-O3-2 + H2O --> R-OH + HO-P-O3-2. Acid phosphatases are produced by erythrocytes, the liver, kidney, spleen, and prostate gland. The enzyme from the prostate gland is clinically important because an increased activity in the blood is frequently an indication of cancer of the prostate gland. The phosphatase from the prostate gland is strongly inhibited by tartrate ion, but acid phosphatases from other tissues are not. How can this information be used to develop a commercial specific procedure for measuring the activity of the acid phosphatase of the prostate gland in human blood serum? * 1. Prostate cancer cannot be diagnosed biochemically. 2. Use tartrate to inhibit phosphatase from prostate gland and then subtract the results from the total serum enzyme activities to get an…arrow_forward5) In an experiment to investigate the inhibition of the enzyme-glucosidase the following data for the rates of reaction with glucopyranoside for various substrate concentrations was obtained. By constructing a Leaver-Burk plot, determine the value of the Michaelis constant. [S]/ (10-6 mol dm-3) v/ (10-3 mol dm-3 s-1) 1.00 2.00 3.00 4.00 16.7 33.3 41.1 49.8arrow_forward
- Intracellular concentrations in resting muscle are as follows: fructose- 6-phosphate, 1.0 mM; fructose-1,6-bisphosphate, 10 mM; AMP, 0.1 mM; ADP, 0.5 mM; ATP, 5 mM; and P, 10 mM. Is the phosphofructokinase reac- tion in muscle more or less exergonic than under standard conditions? By how much?arrow_forwardIntramitochondrial ATP concentrations are about 5 mM, and phos- phate concentration is about 10 mM. If ADP is five times more abundant than AMP, calculate the molar concentrations of ADP and AMP at an energy charge of 0.85. Calculate AG for ATP hydrolysis at 37 °C under these conditions. The energy charge is the concentra- tion of ATP plus half the concentration of ADP divided by the total adenine nucleotide concentration: [ATP] + 1/2[ADP] [ATP] + [ADP] + [AMP]arrow_forwardThe isomerization of dihydroxyacetone phosphate (DHAP) to glyceraldehyde 3-phosphate (GAP) is catalyzed by triose phosphate isomerase. In the cell, the concentration ratio of DHAP/GAP = 5.5. Calculate [DHAP] (in M) when [GAP] = 0.00002arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning

Biochemistry
Biochemistry
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Cengage Learning