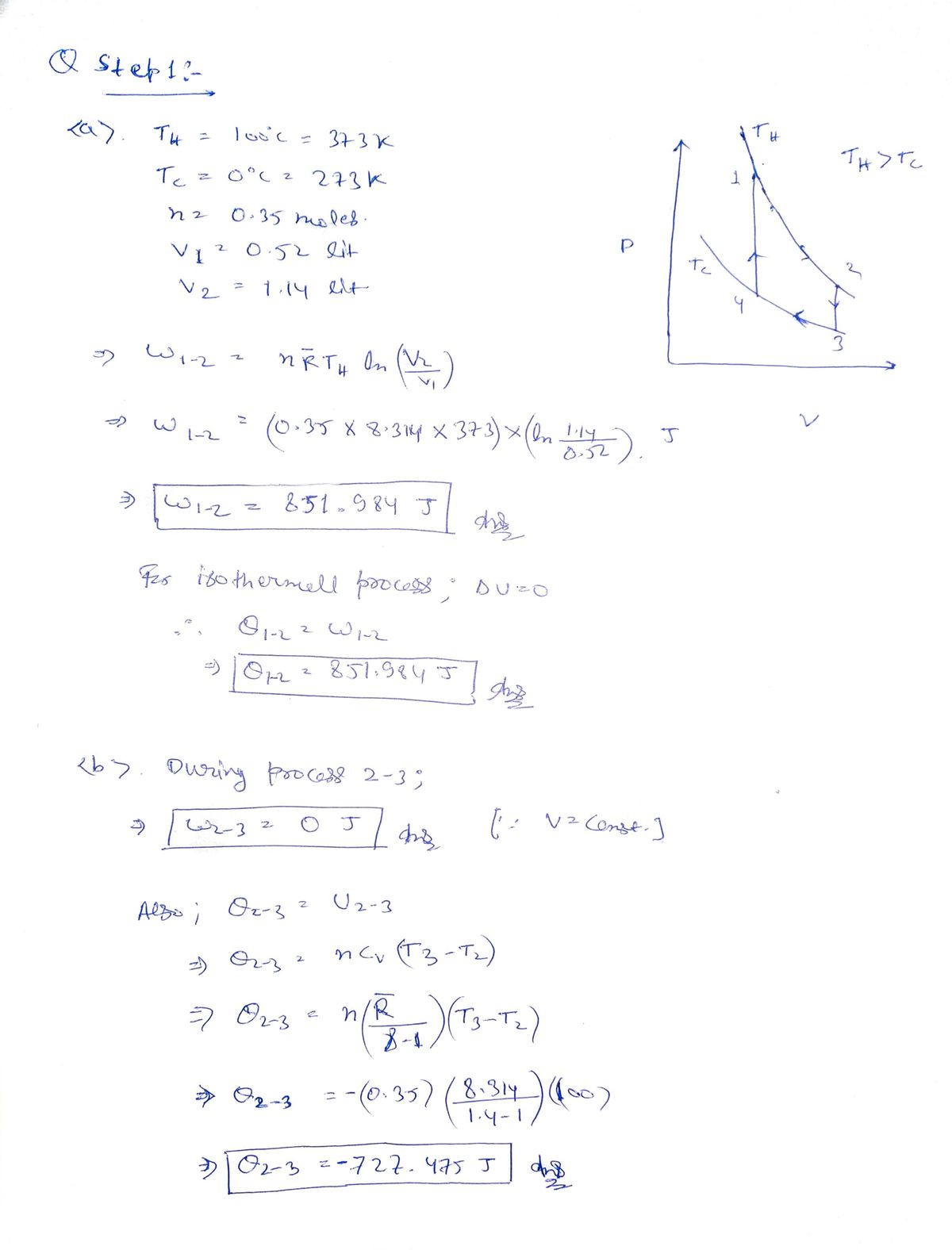
Red pistoln boiling water bath at TH = 100°C. The volume of the piston is 0.52 liters. This point is labeled as 1 in the diagram below. P. Tt > Tc TH Te 3 V V2 (a) The piston is unlocked and the gas is allowed to expand to a volume of 1.14 liters, moving the system from point 1 to point 2. What is the heat flow? What is the work done by the gas? (b) The piston is locked and moved to an ice bath at Tc = 0°C, moving the system from point 2 to point 3. What is the heat flow? What is the work done by the gas? (c) The piston is unlocked to move the system from point 3 to point 4. What is the heat flow? What is the work done by the gas? (a) To move the system from point 4 to point 1, the cylinder is locked and the gas is
Red pistoln boiling water bath at TH = 100°C. The volume of the piston is 0.52 liters. This point is labeled as 1 in the diagram below. P. Tt > Tc TH Te 3 V V2 (a) The piston is unlocked and the gas is allowed to expand to a volume of 1.14 liters, moving the system from point 1 to point 2. What is the heat flow? What is the work done by the gas? (b) The piston is locked and moved to an ice bath at Tc = 0°C, moving the system from point 2 to point 3. What is the heat flow? What is the work done by the gas? (c) The piston is unlocked to move the system from point 3 to point 4. What is the heat flow? What is the work done by the gas? (a) To move the system from point 4 to point 1, the cylinder is locked and the gas is
Principles of Heat Transfer (Activate Learning with these NEW titles from Engineering!)
8th Edition
ISBN:9781305387102
Author:Kreith, Frank; Manglik, Raj M.
Publisher:Kreith, Frank; Manglik, Raj M.
Chapter2: Steady Heat Conduction
Section: Chapter Questions
Problem 2.35P
Related questions
Question

Transcribed Image Text:1. An uninsulated cylinder with a locked piston containing 0.35 moles of gas is placed in a
boiling water bath at TH = 100°C. The volume of the piston is 0.52 liters. This point is
labeled as 1 in the diagram below.
TH> Tc
To
Vi
V2
(a) The piston is unlocked and the gas is allowed to expand to a volume of 1.14 liters,
moving the system from point 1 to point 2. What is the heat flow? What is the work
done by the gas?
(b) The piston is locked and moved to an ice bath at Tc = 0°C, moving the system from
point 2 to point 3. What is the heat flow? What is the work done by the gas?
(c) The piston is unlocked to move the system from point 3 to point 4. What is the heat
flow? What is the work done by the gas?
(d) To move the system from point 4 to point 1, the cylinder is locked and the is
gas
returned to the boiling water bath. What is the heat flow? What is the work done by
the gas?
(e) This heat engine is called a Stirling Cycle. What is its efficiency?
(f) If a Carnot engine were operated using the same boiling water bath and ice bath as
the hot and cold reservoirs, what would its efficiency be?
27
3.
Expert Solution
Step 1
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, mechanical-engineering and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning

Principles of Heat Transfer (Activate Learning wi…
Mechanical Engineering
ISBN:
9781305387102
Author:
Kreith, Frank; Manglik, Raj M.
Publisher:
Cengage Learning