(+)-Tartaric acid has a specific rotation of +12.0°. Calculate the specific rotation of a mixture of 68% (+)-tartaric acid and 32% (-)-tartaric acid.
Q: The answer is C.. please solve with details so I can understand 1. Find the rate law of the…
A:
Q: The equilibrium constant, Kc, for the following reaction is 5.10 × 10-6 at 548 K. NH4Cl(s) NH3(g) +…
A: The question is based on the concept of chemical equilibrium. we need to calculate the equilibrium…
Q: which of the 5 characteristics of gas will generally deviate
A: We are to discuss which of the five characteristics of gas will deviate under applied conditions.…
Q: b. didaction the following organic reactions, as substituto Identify elimmation, or addition Brand…
A: The mechanism of given reaction can be explained as:
Q: Calculate the standard enthalpy change for the following reaction at 25 °C. C₂Hg (g) + 5O₂(g) → 3…
A: The balanced equation is C3H8 (g) + 5 O2 (g) --------> 3 CO2 (g) + 4 H2O (g). ΔHrxn = ? Standard…
Q: The rate of disappearance of HBr in the gas phase reaction 2HBr(g) → H2(g) +…
A:
Q: What set rigid solids apart from amorphous solids, like glass?
A: Rigid solids and amorphous solids are two broad categories of solids that differ in their molecular…
Q: 1b.) You also wish to make 200mL of a 0.02 mg/mL solution. (Hint: NEW V₂. and C₂). Calculate the two…
A: We have to prepare 200 mL of 0.02 mg/mL solution from the stock solution. So, the initial…
Q: How many kinds of chemically non-equivalent hydrogens are there in each of the following compounds?…
A: We have to determine the number of chemically non-equivalent hydrogens present on the given…
Q: 5. The reaction shown below is balanced, use it to answer the following questions. 4Fe(s) + 302(g)…
A: Since you have mentioned for parts : a, c and d only. We are providing you a solution for these.
Q: A sample of 3.10 mol of helium is confined at low pressure in a volume at a temperature of 80 °C.…
A: Given , moles of helium in sample = 3.10 mol Temperature , T = 80°C
Q: Identify the conjugate bases corresponding to the following acids: a) [Co(NH3)(OH₂)]³+ b) HSO4 c)…
A: Conjugate acid-base pair is the pair of compounds that differ by a proton. A proton is added to…
Q: Classify each of the molecules below. OH O primary alcohol Osecondary alcohol C tertiary alcohol C…
A: There are three types of alcohol on the basis of carbon that is directly attached with oxygen :…
Q: Draw the product of the reaction between a ketone and an alcohol. Include all hydrogen atoms in the…
A:
Q: When is anything in equilibrium? When will we know if things are balanced
A: In chemistry, equilibrium refers to a state in which the rate of the forward reaction is equal to…
Q: do you think that the elements X and Y are metals or nonmetals lastly, In terms of the activity…
A: This question belongs to Electrochemistry.
Q: Calculate the hydrogen ion concentration of a basic solution with a pH of 11.83.
A: Consider the given information as follows; pH = 11.83 Hydrogen ion concentration H+ =?
Q: For the following reactions, look at the starting materials and try to find a comparable substance…
A: The question is based on concept of chemical equilibrium and acid base. we need to identify the…
Q: Fill in the name and empirical formula of each ionic compound that could be formed from the ions in…
A: The formula of ionic compounds from the ions can be determined as follows:
Q: 5. Carbon tetrachloride may be produced via the reaction between methane and chlorine gases: CH₁ (g)…
A:
Q: Convert the concentration of 0.700 M Na₂SO₄ to g/mL
A: Given, concentration = 0.700 M (mol/L) we know, molar mass of sodium sulfate (Na2SO4) = 142.04…
Q: A buffer solution was prepared by dissolving 0.04 mol Na2HPO4(s) and 0.02 mol NaH2PO4(s) in water…
A: In this buffer solution, the buffering molecules are the conjugate acid-base pair of NaH2PO4 and…
Q: 4 5 warmth/ neat (d) Word Equation: Cloudy Precipitate/ d) Word Equation: Sodium Hydroxide +…
A: Given that, 4.Sodium hydroxide + hydrochloric acid (heat) 5.Barium chloride + Sulphuric acid(Claudy…
Q: didention the following organic reactions as substitution elimination, or addition of from arrows to…
A: This question belong to reaction mechanism of organic compounds. Substitution Reaction: one…
Q: The following reactions all have K > 1. 1) HF (aq) + HCOO- (aq) = F- (aq) + HCOOH (aq) 2) HCgHyO4…
A: The Bronsted-Lowry acid is the subtance which donates protons and form corresponding conjugate…
Q: A(aq) + B(aq) C(aq) 1. Write the mass action expression for Kc, the equilibrium constant. 2. If the…
A: This question belong to Chemical Equilibrium. This question will be solve with the help of…
Q: Some patterns of electron configuration are listed below. In each case, [G] stands for a noble-gas…
A: The question is based on periodic table. we need to identify the element from its configuration and…
Q: When calculating enthalpy of reaction values, we use the "products minus reactants" mathematical…
A: The enthalpy of formation is the enthalpy change that occurs when one mole of a compound is formed…
Q: A reaction where A ,B, and C react to form products is zero order in A, one-half order in b, and…
A: reaction where A ,B, and C react to form products is zero order in A, one-half order in b, and…
Q: For the equation H2O2---> 2H2O + O2 Calculate the average rate of the reaction between 10 and 20…
A: This question belong to chemical kinetics. Average rate of reaction = [Cf - Ci ]/[tf - ti ]
Q: Consider the equilibrium system described by the chemical reaction below. At equilibrium, a sample…
A: This can be explained as :
Q: The reaction below has rate constant 2.50 x 10 -1 min -1 and obeys the rate law: rate = k…
A:
Q: Cerium-141 is a radioisotope used to assess blood flow through the heart. What new radioisotope is…
A:
Q: Classify each of the following as either a substitution, elimination, or addition reaction. OH…
A:
Q: 38°F Cloudy Two sets of ionizations are shown in the tables below. Complete the tables by ordering…
A: Ionisation energy is the amount of energy needed to remove electrons from outermost shell of…
Q: Test tube 1 contain hexane, test tube 2 contains fluorene, test tube 3 contains Fluorenone On the…
A: The Rf (retention factor) value in chromatography is related to the polarity of the compound. The Rf…
Q: AS or additions Draw electron arrows to show how the bends break and Vintermediates of carbon. form…
A: There are so many types of reactions. But this type of reaction is an different one. This is an…
Q: Part I. Data Table mass of empty Styrofoam cup and lid mass of cup, lid and cool water temperature…
A: Percentage error : To calculate percentage error, subtract the true value from the measured value,…
Q: Estimate the concentration of sucrose after 175 min.
A: The concentration of sucrose after 175 min can be calculated as:
Q: A student performs an experiment to determine the density of a sugar solution. She obtains the…
A: Given that, Experimentally measured values of density of a sugar solution by a student are 1.11…
Q: A mixed aldol condensation in which a ketone reacts with an enolizable aldehyde or ketone. O Michael…
A: Given Mixed aldol condensation reaction
Q: The 2021 Nobel Prize in Chemistry was awarded to two organic chemistry scientists. Explain in your…
A: The 2021 Nobel Prize in Chemistry was awarded jointly to Benjamin List and David W.C. MacMillan for…
Q: Is this a polar molecule? :F-F: Yes No Is this a polar molecule? H-Br: O Yes O No
A: Polar compound is one in which electrons are share unequally between two elements. In polar…
Q: Fill in the name and empirical formula of each ionic compound that could be formed from the ions in…
A: The question is based on the concept of chemical composition. we need to write the formula of the…
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a…
A: Answer:- This question is answered by using the simple concept of calculation of equilibrium…
Q: Attached is the NMR data sheet for 4-cyclohexene-cis-dicarboxylic anhydride. How do I interpret it…
A: Integration is generally the number of one type of proton due to which signal arises (in the above…
Q: A glucose solution is frequently used as an intravenous (IV) solution to supply hydration and/or…
A:
Q: Fix any errors in these proposed electron configurations. number of electrons in atom 14 8 23…
A:
Q: Two or more substances in variable proportions, where the composition is constant throughout are a…
A: A compound is a chemical species that is formed when two or more atoms join together chemically,…
Q: The condensed cell diagram for the reaction occurring in silver–zinc button batteries is (-) Zn(s)…
A: The cell diagram for the reaction is (-) Zn(s) | ZnO(s) | KOH(aq) || Ag2O(s) | Ag(s) (+) The…

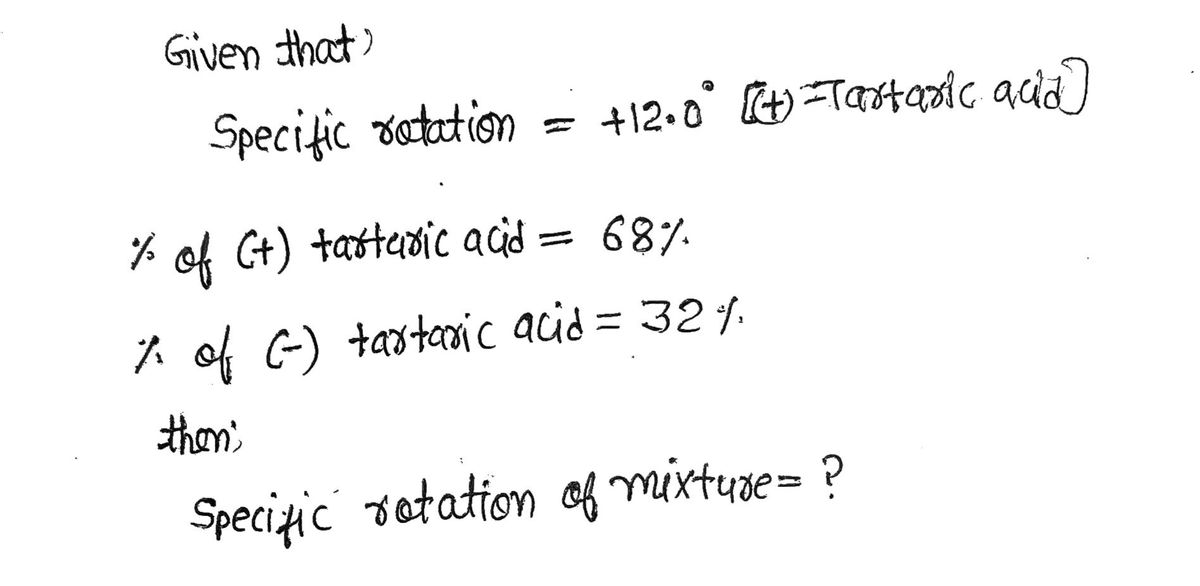
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- a) Draw the structure of the compound: (R) -1,1,2-trimethylcyclohexane. b) (+)-Oxalic acid has a specific rotation of +14.0°. Calculate the specific rotation of a mixture of 58% (+)-Oxalic acid and 42% (-)-Oxalic acid.CH-ST53 The specific rotation of dextrorotatory tartaric acid is +12.7 degrees. A mixture of dextrorotatory and levorotatory tartaric acid has a specific rotation of -6.35 degrees. What is the proportion of each compound in the mixture? O A. 25% of the dextrorotatory tartaric acid and 75% of the levorotatory tartaric acid O B. 33% of the dextrorotatory tartaric acid and 67% of the levorotatory tartaric acid OC. 50% of the dextrorotatory tartaric acid and 50% of the levorotatory tartaric acid O D.75% of the dextrorotatory tartaric acid and 25% of the levorotatory tartaric acid(+) Chloramphenicol is a powerful antibiotic isolated in 1949 from the Streptomyces venezuelae bacterium. Its specific rotation is +18.6°. What is the composition of a mixture of (+) and (-) chloramphenicol whose observed specific rotation is +14.88° ? Select one: A. 60% (+) chloramphenicol and 40 % (-) chloramphenicol B. 70% (+) chloramphenicol and 30 % (-) chloramphenicol C. 90% (+) chloramphenicol and 10 % (-) chloramphenicol D. 80% (+) chloramphenicol and 20 % (-) chloramphenicol
- The pure D-form of a compound has a specific rotation (D line of sodium, 20° C) of +24°, whereas the pure L- form of the compound has a specific rotation (D line of sodium, 20° C) of -24°. If a scientist has a sample of this compound that is known to be 78% pure D-form (and, therefore, 100% - 78% pure L-form), what will be the observed rotation of this sample? Enter your answer to the nearest hundredth. The units are assumed to be degrees. 13.441) The specific rotation of pure (+)-carvone gives a specific rotation of +61.0°. Is this compound dextrorotatory or levorotatory? Is the compound R or S? 2) A sample of carvone was isolated from spearmint oil. The sample gave a specific rotation of 30.5°. a. What enantiomer is the major enantiomer present in spearmint oil? b. In the sample, what is % ee of the major enantiomer? c. In the sample, what is the total % of the major enantiomer? d. In the sample, what is the total % of the minor enantiomer? e. What part of the sample constitutes the racemic mixture of the enantiomers?13. If the proton-pump inhibitor, (S)-omeprazole, has a specific rotation (rotation of plane polarized light) of -155°, what should the specific rotation of (R)-omeprazole be? a) -155° b) 0° c) +155° d) It can only be determined experimentally
- A sample mixture containing the made-up enantiomers (R)-(+)-Nenamide and (S)-(-)-Nenamide has an apparent specific rotation of +6.50° at 25 °C. This sample has an enantiomeric excess (EE) of 43.0%. Provide answers to the following questions : 1.Which enantiomer is in excess in the sample? 2.What is the specific rotation of (S)-(-)-Nenamide?An attempt at synthesizing a certain optically-active compound resulted in a mixture of its enantiomers. The mixture had an observed specific rotation of +16.0°. If it is known that the specific rotation of the R enantiomer is -33.3°, determine the percentage of each isomer in the mixture. R enantiomer: S enantiomer: % %The specific rotation of (S)-carvone (at 20 degrees) is +61. A chemist prepared a mixture of (R)-carvone and its enantiomer, and this mixture had an observed rotation of -55. (a) What is the specific rotation of (R)-carvone at 20 degrees?
- Naproxen, a nonsteroidal anti-inflammatory drug that is the active ingredient in Aleve, has a specific rotation of +66. One commercial preparation results in a mixture with a 97% enantiomeric excess. a. Does naproxen have the R or the S configuration? b. What percent of each enantiomer is obtained from the commercial preparation?C 23. a) The observed rotation for a solution containing the R and S enetaiomers a of a sample was +17.6. An enantiomerically pure sample of the S enantiomer has a specific rotation of + 22.3°. What is the % enantiomeric excess of sample A? b)A pure sample of the R-enantiomer of a compound has a specific rotation of -15°. A solution containing 0.6 g/mL of a mixture of enantiomers rotates plane polarized light by -3° in a 1 dm polarimeter. What is the enantiomeric excess (%ee) of the mixture? 9H. В A (a) Assign the configuration (R or S) of each chirality center (labeled A to C) in coibacin B. (b) Identify the number of possible stereoisomers for this compound, assuming that the geometry of the alkenes are fixed. Choices are given below and write the CAPITAL LETTER of your choice. А. 2 В. 4 C. 8 D. 16

