Organic And Biological Chemistry
7th Edition
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:STOKER, H. Stephen (howard Stephen)
Chapter3: Alcohols, Phenols And Ethers
Section: Chapter Questions
Problem 3.79EP
Related questions
Question
100%
Help

Transcribed Image Text:Question 9 of 40
Submit
What is the correct IUPAC name for the
compound shown here?
6-3- 5- 2-
sec- iso tert-
pentan heptan methyl propyl hexan ethyl
oate one al ol oic acid

Transcribed Image Text:Submit
Question 10 of 40
What is the correct IUPAC name for the
compound shown here?
CH3
||
H-C-CH2-CH-CH3
3-6-5-) 4- 2-
tri di
butan ethyl methyl pentan propyl propan
al one ol)oate oic acid
O=
Expert Solution
Step 1
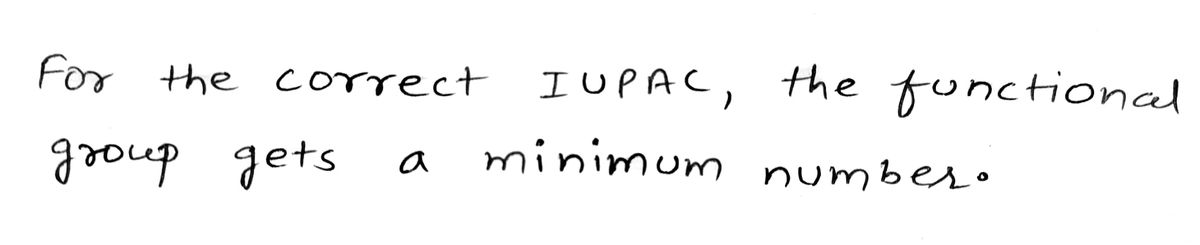
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning