
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
4th Edition
ISBN: 9781119776741
Author: Klein
Publisher: WILEY CONS
expand_more
expand_more
format_list_bulleted
Question
Chapter 10.6, Problem 6LTS
Interpretation Introduction
Interpretation:
The stereochemical outcome of radical bromination of the following
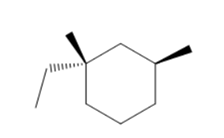
Concept Introduction :
As a branch of chemistry, stereochemistry focuses on modifying and studying the relative spatial arrangement of the atoms that constitute molecules.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For following substituted benzenes:
[1] C6H5Br; [2] C6H5CN; [3] C6H5OCOCH3:
Does the substituent activate or deactivate the benzene ring inelectrophilic aromatic substitution?
The sex attractant by which the female housefly attracts the male has the molecular formula C23H46. Catalytic hydrogenation yields an alkane of molecular formula C23H48. Ozonolysis yieldsWhat is the structure of the housefly sex attractant?
Alloocimene is a hydrocarbon found in turpentine. It has the molecular formula C₁0H16 and a UV absorption maximum at 290
nm. On hydrogenation with a palladium catalyst, 2,6-dimethyloctane is obtained. Ozonolysis of alloocimene, followed by
treatment with zinc and acetic acid, produces the following four fragments:
||
CH3C-CH
i
O
||
||
CH3CCH3 HC-CH
Propose a structure for alloocimene.
O
||
CH3CH
Chapter 10 Solutions
EBK ORGANIC CHEMISTRY-PRINT COMPANION (
Knowledge Booster
Similar questions
- Identify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forwardWhich of the following reactions results in Markovnikov addition of H2O to an alkene?arrow_forwardHow many alkenes yield 2,3−dimethylbutane on catalytic hydrogenation?arrow_forward
- Bromine reacts with alkenes in methanol according to the equation: - When this reaction was carried out with 4-tert-butylcyclohexene, only one isomer was formed with the molecular formula C12H23BrO (80% yield). Which of the following is the structure more reasonable for this compound?. Explain your reasoning through acorresponding mechanism.arrow_forwardPlease give the main substitution product for each of the following reactions, and indicate the dominant mechanism: (a) 1-bromopropane + NaOCH3 → (b) 3-bromo-3-methylpentane + NaOC2H5 →arrow_forwardPredict the products of additions, oxidations, reductions, and cleavages of alkynes, including the orientation of the reaction (regiochemistry) and the stereochemistry.arrow_forward
- Name the following compounds. Include stereochemistry where appropriate:arrow_forwardThe reaction of (S)-2-bromopentane with potassium cyanide to yield 2-methylpentanenitrile (2-cyanopentane) occurs due to a nucleophilic substitution pathway. The reaction is 100% stereospecific. Please explain what this observation tells about the mechanism of the reaction.arrow_forwardwhich of the following alkenes will most likely undergo a hydride shift when reacting with HCl?arrow_forward
- which among the following alkenes will most likely undergo a hydride shift when reacting with HCl?arrow_forwardFor each reaction below, provide the structural formula for the reactants and the predicted products. Indicate which mechanism the reaction will follow and use curved arrow formalism to illustrate the predicted reaction mechanism. t-butylalcohol + NaBr ?arrow_forwardWhat is the major product of the following reactions? Disregard stereoisomers:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning