
Write a structure for each of the following
- a. N,N – dimethylhexanamide
- b. 3,3 – dimethylhexanamide
- c. propionamide
- d. sodium acetate
- e. butyric anhydride
- f. 3 – methylbutanenitrile
(a)
Interpretation:
The strucutre for the given name of the compound has to be drawn.
Concept introduction:
The structure of the organic compound and its name are closely related to each other. The structure of the compound can be drawn if the name is given and vice-versa. The name of an organic compounds tells about the number of carbons present in the compound which is necessary so as to draw the carbon skeleton in the structure. The suffix of the name of the compound provides information about the functional group present in the compound.
Answer to Problem 31P
The structure of
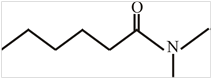
Explanation of Solution
The structure of the compound can be drawn if the name is given and vice-versa. The name of the compound is
Thus, the structure is drawn as,

(b)
Interpretation:
The strucutre for the given name of the compound has to be drawn.
Concept introduction:
The structure of the organic compound and its name are closely related to each other. The structure of the compound can be drawn if the name is given and vice-versa. The name of an organic compounds tells about the number of carbons present in the compound which is necessary so as to draw the carbon skeleton in the structure. The suffix of the name of the compound provides information about the functional group present in the compound.
Answer to Problem 31P
The structure of
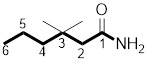
Explanation of Solution
The structure of the compound can be drawn if the name is given and vice-versa. The name of the compound is
Thus, the structure is drawn as,
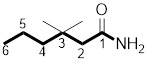
(c)
Interpretation:
The strucutre for the given name of the compound has to be drawn.
Concept introduction:
The structure of the organic compound and its name are closely related to each other. The structure of the compound can be drawn if the name is given and vice-versa. The name of an organic compounds tells about the number of carbons present in the compound which is necessary so as to draw the carbon skeleton in the structure. The suffix of the name of the compound provides information about the functional group present in the compound.
Answer to Problem 31P
The structure of
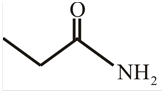
Explanation of Solution
The structure of the compound can be drawn if the name is given and vice-versa. The name of the compound is
Thus, the structure is drawn as,

(d)
Interpretation:
The strucutre for the given name of the compound has to be drawn.
Concept introduction:
The structure of the organic compound and its name are closely related to each other. The structure of the compound can be drawn if the name is given and vice-versa. The name of an organic compounds tells about the number of carbons present in the compound which is necessary so as to draw the carbon skeleton in the structure. The suffix of the name of the compound provides information about the functional group present in the compound.
Answer to Problem 31P
The structure of
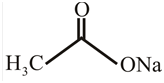
Explanation of Solution
The structure of the compound can be drawn if the name is given and vice-versa. The name of the compound is
Thus, the structure is drawn as,

(e)
Interpretation:
The strucutre for the given name of the compound has to be drawn.
Concept introduction:
The structure of the organic compound and its name are closely related to each other. The structure of the compound can be drawn if the name is given and vice-versa. The name of an organic compounds tells about the number of carbons present in the compound which is necessary so as to draw the carbon skeleton in the structure. The suffix of the name of the compound provides information about the functional group present in the compound.
Answer to Problem 31P
The structure of
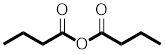
Explanation of Solution
The structure of the compound can be drawn if the name is given and vice-versa. The name of the compound is
The suffix is ‘anhydride’ indicating the presence of an acid anhydride group in which the
Thus, the structure is drawn as,

(f)
Interpretation:
The strucutre for the given name of the compound has to be drawn.
To draw the structure of the given compounds.
Concept introduction:
The structure of the organic compound and its name are closely related to each other. The structure of the compound can be drawn if the name is given and vice-versa. The name of an organic compounds tells about the number of carbons present in the compound which is necessary so as to draw the carbon skeleton in the structure. The suffix of the name of the compound provides information about the functional group present in the compound.
Answer to Problem 31P
The structure of 3-methylbutanenitrile is:
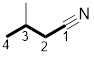
Explanation of Solution
The structure of the compound can be drawn if the name is given and vice-versa. The name of the compound is 3-methylbutanenitrile. It indicates that four carbons are present in the carbon skeleton of the compound. The suffix is ‘nitrile’ indicating the presence of a nitrile functional group. There is a methyl group on the third carbon in the chain. The numbering of carbons starts from the functional group side.
Thus, the structure is drawn as,

Want to see more full solutions like this?
Chapter 11 Solutions
Essential Organic Chemistry, Global Edition
- Which compound has the highest boiling point? A. methoxyethane B. trimethylamine C. ethylmethylamine D. propylamine E. isopropylaminearrow_forwardDraw the structures of the following hydrocarbon derivatives. a. Butyl heptanoate b. 3-chloro-4-ethylbenzoic acid c. N,N-diethylpent-3-en-1-amine d. 4-methoxyhept-2-ene e. 3-ethyl-N,2-dimethylhexan-1-amidearrow_forward1. what grouo does the ff organic compound belong? a. ketone b. ether c. cyloalkane d. esther 2. what group does the ff organic compound belong? a. amide b. azo c. nitrile d. amine 3. what is the priority functional group of the ff organic compound? a. carboxyl b. hydroxyl c. carbonyl d. hydroxidearrow_forward
- Draw structural diagrams for the following molecules a. N-ethyl-2-methyl-butan-1-amine b. 3,4-dihydroxy-4-fluoro-pentanoic acidarrow_forward1. Draw a simple organic molecule that contain the following functional groups. a. Cycloalkane d. Ester b. Amine c. Ether e. Aldehyde f. Ketonearrow_forwardBased on the image attached, what is the name of the compound? A. 3-chloro-2-ethylpyrimidine B. 3-chloro-2-ethyl-1,4-diazacyclohexane C. 3-chloro-2-ethylcyclohexan-1,4-diamine D. 2-chloro-3-ethyl-1,4-diazacyclohexanearrow_forward
- Amide formation involves which two functional groups? A.carboxylic acid and alcohol B.alcohol and ketone C.carboxylic acid and amine D.amine and alcoholarrow_forwardThe dehydration of an alcohol produces which organic functional group and steam? a. alkyne b. aldehyde c. alkene d. ketone e. carboxylic acid and an alcoholarrow_forward2. Draw the following molecules 2-methyl-4-propyloctane a. b. 2-heptanone c. Methylbutyletherarrow_forward
- 4. A functional group with a carbonyl group functionality is _________________.A. Alcohol B. Amine C. Ester D. Ketonearrow_forwardWrite the condensed formula for each of the following compounds.a. 3-Methylnonanal c. 4-Fluorohexanalb. b-Bromovaleraldehyde d. a,b-Dimethylbutyraldehydearrow_forward41. Draw structures for the following names. a. methyl pentanoate b. heptyl methanoate c. butyl ethanoate d. propyl octanoate e. ethyl 3,3-dimethylbutanoate f. octyl ethanoate (found in oranges)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





