
Concept explainers
(a)
Interpretation:
The difference between ionic compound and molecular compound in scientific terms is to be stated.
Concept introduction:
An ionic bond is formed by the transfer of electrons, which is also known as electrovalent bond. The compound which are composed of molecules contain more than one type of atom are called molecular compounds.
Answer to Problem 41E
An ionic compound is formed by the transfer of electrons, while a molecular compounds are composed of molecules contain more than one type of atom.
Explanation of Solution
The compound which contains ionic bond between two or more atoms is known as ionic compound. Ionic bonds are formed between electropositive and electronegative elements, which have larger electronegativity difference. Sodium chloride is an example of ionic compound, which is shown below.
![]()
Figure 1
The compound which is composed of molecules containing more than one type of atom is called molecular compounds. Methane is an example of molecular compound, which is shown below.
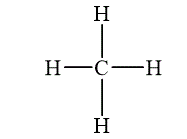
Figure 2
An ionic compound is formed by the transfer of electrons, while a molecular compounds are composed of molecules contain more than one type of atom.
(b)
Interpretation:
The difference between ionic bond and covalent bond in scientific terms is to be stated.
Concept introduction:
An ionic bond is formed by the transfer of electrons, which is also known as electrovalent bond. Ionic bonds are formed between electropositive and electronegative elements. A covalent bond is formed by the mutual sharing of electrons. The number of electrons contributed by each atom is known as covalence of the atom. The covalent bond is stronger than ionic bonds because of sharing of the electrons.
Answer to Problem 41E
An ionic bond is formed by the transfer of electrons, while a covalent bond is formed by the mutual sharing of electrons.
Explanation of Solution
An ionic bond is formed by the transfer of electrons, which is also known as electrovalent bond. Ionic bonds are formed between electropositive and electronegative elements. Sodium chloride is an example that includes ionic bonding, which is shown below.
![]()
Figure 1
A covalent bond is formed by the mutual sharing of the electrons. The number of electrons contributed by each atom is known as covalence of the atom. Hydrogen chloride is an example that describes covalent bonding, which is shown below.
![]()
Figure 3
An ionic bond is formed by the transfer of electrons, while a covalent bond is formed by the mutual sharing of electrons.
(c)
Interpretation:
The difference between lone pair and bonding pair in scientific terms is to be stated.
Concept introduction:
Lone pair of electrons is the type of electrons, which do not participate in the bond formation. Bonding pair of electrons are type of electrons, which participate in the bond formation and present between the bonded atoms.
Answer to Problem 41E
Lone pair of electrons are electrons, which do not take participate in the bond formation, while bonding pair of electron are electrons, which participate in the bond formation.
Explanation of Solution
Lone pair of electrons are electrons, which do not participate in the bond formation. Lone pair of electrons are localized on the atom. Bonding pair of electron are electrons, which participate in the bond formation and present between the bonded atoms. The example, which shows lone pair and bonding pair is shown below.
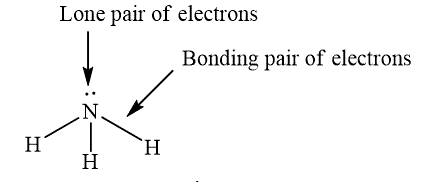
Figure 4
Loan pair of electrons are electrons, which do not participate in the bond formation, while bonding pair of electron are electrons, which participate in the bond formation.
(d)
Interpretation:
The difference between nonpolar bond and polar bond in scientific terms is to be stated.
Concept introduction:
Polar and non polar bonds are the type of covalent bond. A polar bond is formed between atoms of different elements, having some value of electronegativity difference. A non polar bond is formed between atoms of same elements.
Answer to Problem 41E
A polar bond is formed between atoms of different elements, while a non polar bond is formed between atoms of same elements.
Explanation of Solution
A polar bond is formed between atoms of different elements, having some value of electronegative difference. A non polar bond is formed between atoms of same elements.
The example of polar and non polar bond is shown below.
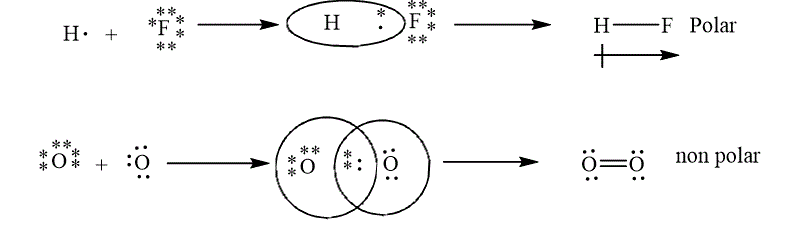
Figure 5
A polar bond is formed between atoms of different elements, while a non polar bond is formed between atoms of same elements.
(e)
Interpretation:
The difference among single, double, triple, and multiple bonds in scientific terms is to be stated.
Concept introduction:
If two atoms are bonded with the sharing of one pair of electron, such a bond is called as single bond. If two atoms are bonded by the sharing of two pairs of electrons, such a bond is called as double bond.
Answer to Problem 41E
Single bond is formed by the sharing of one pair of electron between two atoms. Double bond is formed by the sharing of two pair of electron between two atoms. Triple bond is formed by the sharing of three pair of electron between two atoms. Multiple bonds is the general term that includes double and triple bonds.
Explanation of Solution
If two atoms are bonded with the sharing of one pair of electron, such a bond is called as single bond. The example of a single bond is shown as below.
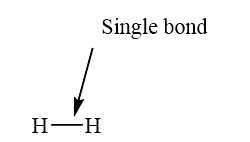
Figure 6
If two atoms are bonded by the sharing of two pairs of electrons, such a bond is called as double bond. The example of double bond is shown as below.
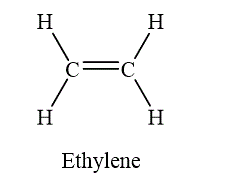
Figure 7
If two atoms are bonded by three pairs of electrons, such bond is called as triple bonds. The example of triple bond is shown as below.
![]()
Figure 8
Multiple bonds is a general term that includes double and triple bonds. The example of multiple bond is shown as below.
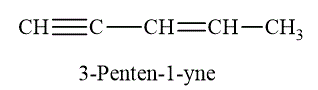
Figure 9
Single bond is formed by the sharing of one pair of electron between two atoms. Double bond is formed by the sharing of two pair of electron between two atoms. Triple bond is formed by the sharing of three pair of electron between two atoms. Multiple bonds is a general term that includes double and triple bonds.
Want to see more full solutions like this?
Chapter 12 Solutions
Introductory Chemistry: An Active Learning Approach
- The molecule is HFarrow_forwardIs it correct to say that a covalent bond is the electrostatic attraction between negative shared electrons and the positive nuclei of the two adjacent atomsarrow_forwardWhich of these describes the formation of ionic and covalent bonds? a.) electrons are transferred between atoms to form ions in ionic bonds b.) atoms forming a covalent bond have different electron affinities c.) covalent bonds do not share electrons between atoms d.) electrons are shared equally between atoms in ionic bondsarrow_forward
- 2. What is the term for a compound composed of two nonmetal atoms held together by a covalent bond? A) diatomic formula unit B) nonmetal unit C) nonpolar molecule D) polar molecule E) diatomic moleculearrow_forwardState the relationship among an atom, molecule, and compound.arrow_forwardIn chemical bonding, metals tend to lose electrons when forming an ionic compound. What is the connection with the electronegativity of metals?arrow_forward
- In a covalent bond between a oxygen atom and a sulfur atom, which of the atoms has a partial positive charge?arrow_forwardPrelaboratory Questions Bathay Crant lame Laboratory Section 1. a) When magnesium forms a compound with oxygen, electrons are transferred from the atom to the atom. b) What type of bonding exists between the magnesium and oxygen atoms? c) Indicate the electrical charge on each of the ions and the total electrical charge on the compound. on?arrow_forward1. In the blank beside the following statement, write "" if the statement applies to ionic bonds and "C" if the statement applies to covalent bonds. (a) form between atoms (b) are created by the sharing of pairs of valence electrons between entities (c) hold together the atoms within molecules (d) are created by a transfer of valence electrons between entitiesarrow_forward
- 45. Select which of the following are correctly identified. a) b) c) d) Both CO and KBr are polar covalent molecules. Both MgO and HCl are ionic compounds. KBr is an ionic compound and I, is a nonpolar covalent molecule. CO and H2 are nonpolar covalent molecules.arrow_forwardwhat are the bond polarity and molecular polarity for the following?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





