
Concept explainers
You are experimenting with several liquid metal alloys to find a suitable replacement for the mercury used in thermometers. You have attached capillary tubes with a circular cross section and an inside diameter of 0.3 millimeters to reservoirs containing 5 cubic centimeters of each alloy. You mark the position of the liquid in each capillary tube when the temperature is 20 degrees Celsius, systematically change the temperature, and measure the distance the liquid moves in the tube as it expands or contracts with changes in temperature. Note that negative values correspond to contraction of the material due to lower temperatures. The data you collected for four different alloys is shown in the following table.
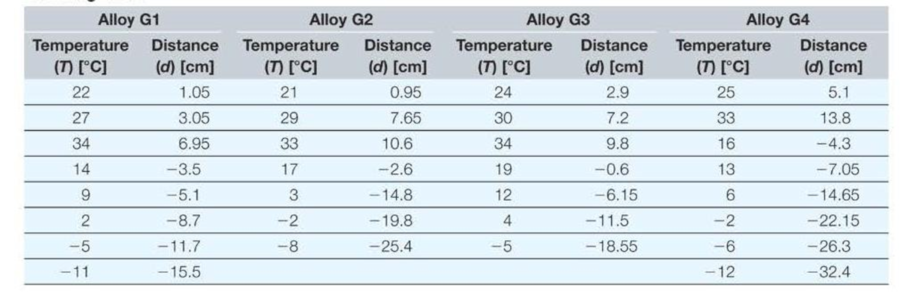
- a. In Excel, create two new columns for each compound to calculate the change in temperature (∆T) relative to 20 °C (for example, 25 °C gives ∆T = 5 °C) and the corresponding change in volume (∆V).
- b. Plot the change in volume versus the change in temperature: fit a linear trendline to each data set.
- c. From the trendline equations, determine the value and units of the coefficient of thermal expansion, β, for each alloy. Note that ∆V = βV∆T, where V is the initial volume.
- d. There is a small constant offset (C) in each trendline equation (∆V = βV∆T + C). What is the physical origin of this constant term? Can it be safely ignored? In other words, is its effect on the determination of β negligible?
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
Thinking Like an Engineer: An Active Learning Approach (4th Edition)
Additional Engineering Textbook Solutions
Mechanics of Materials (10th Edition)
Engineering Mechanics: Dynamics (14th Edition)
Mechanics of Materials
Statics and Mechanics of Materials (5th Edition)
Automotive Technology: Principles, Diagnosis, And Service (6th Edition) (halderman Automotive Series)
Engineering Mechanics: Statics
- The volume in buoyancy formula is the volume of the entire object. Select one: True False Reynold's number identify the laminarity of the fluid. Select one: True False If the velocity in a given location is invariant with time the flow is said to be non-steady. Select one: True O False The viscosity of a fluid is a measure of its resistance to defeormation. Select one: True O False Flow rate is the amount of fluid passing through a section per unit of pressure. Select one: True O Falsearrow_forwardYou drive on 15 to the Bay Area. Midway through the trip your car indicates that the tire pressure is low. The pressure is supposed to be 330 kPa (absolute) but you measure it to be 290 kPa. Due to driving, the temperature of the air in the tires when they are filled is 50 °C. You add air to the desired pressure value for your tires and keep driving. The next morning, the air in the tires has equilibrated to San Francisco morning temperatures of 10 °C, and your car still says the tire pressure is low. Assume the tire volume to be fixed. Take C₂ = 1.005 kJ/kg-K and C₂ = 0.718 kJ/kg-K, T = 132 K, and Pc = 3.77 MPa. (a) In words, describe why the tire pressure is low the next morning, even though you filled up the tires midway through your trip. (b) (c) (d) (e) Sketch a T - v diagram of the process. Determine whether the ideal gas law is appropriate to employ for further analysis in this situation. Your argument should involve numbers. Calculate the pressure of the tires the next morning…arrow_forwardYou are working with an imagined cubical pot of water, 1 meter on each side, sitting on a stove. Heat is applied to the bottom of the pot from the stovetop. The pot has been on the stove for a while, so the stovetop is hot and some heat has already been added to the water. Assume the water at the bottom of the pot is held at 99 degrees C , while the water at the top of the pot is cold, 4 degrees C. Compute the heat delivered by that 1 g of water at 99 °C into the layer of water at the top of the pot, assuming the latter is 1 meter square, 1 cm deep, and 4 °C. Start by solving the calorimetry problem (Section 14-4 in the text) to determine the final temperature of the top layer plus hot 1 gram from below. Then use that to determine how much heat was delivered in the hot waterarrow_forward
- In addition to filling in the blanks below, show all of your work for this problem on paper for later upload. You are cooking dinner for a friend in Denver on a day in which the atmospheric pressure is 0.831 atm. Given that the normal boiling point of water is 100.0 °C and the heat of vaporization of water is 40.66 kJ mol1, compute the temperature of boiling water in Denver. Enter your value in the first box and an appropriate unit of measure in the second box. Aarrow_forwardPLEASE SHOW SOLUION STEP BY STEP WITH UNITS. PLEASE ANSWER IT IN 30 MINS. TOPIC: THERMODYNAMICS - PROCESSES OF GASESarrow_forwardOn an alien planet, the law that expresses the flow of blood through a blood vessel, F, as a function of the radius r of the vessel is f=kr5/2where k is a constant. Certain geological features of this planet produce a gas that changes the radius of blood vessels by 5% at the time of inhalation. What would the corresponding increase in blood flow through the vessel at this time be? Write 1-2 sentences explaining how you found your answer.arrow_forward
- The pressure of a car tire depends on the temperature of the air in the tire. While the air temperature is 25 degrees Celsius, the effective pressure is 250 kilo pascals. If the volume of the tire is 0.025 m³, what is the pressure that will occur when the temperature in the tire reaches 50 degrees Celsius?arrow_forward⦁ A hot air balloon holds 7.40 x 107 liters of gas. In order for the hot air balloon to rise into the sky, it must be heated to 120°C. Once the balloon is up in the air, the air needs to be cooled in order for the balloon to land. If the temperature of the balloon is decreased to 40.0°C, what will the new volume of the gas be? Remember to pay close attention to the units of temperature before beginning your calculations.arrow_forwardConsider an introductory thermodynamics class experiment used to demonstrate phase change phenomena. A beaker of water is heated, and its temperature measured over time to establish the temperature at which boiling occurs. The results, shown in the Figure below, are for three separate tests conducted on different days by different student groups using the same equipment and method. Why might the data from three seemingly identical tests show different results? Temperature (°C) 101 100 99 98 97 96 95 94 1 2 3 4 Time (min) 5 Boiling region 100.3 100.1 99.8 Boiling point results Test 1 (762 mm Hg) Test 2 (754 mm Hg) Test 3 (767 mm Hg) 6 7arrow_forward
- Study the table to the right and then answer these questions by filling in the blank columns in the table.1. Convert the miles per gallon figures in the table tokilometers per liter (kpl).2. How many liters (and how many gallons) of gasolinewould each type of car use annually if it were driven19,300 kilometers (12,000 miles) per year?3. How many kilograms (and how many pounds) of carbon dioxide would be released into the atmosphereannually by each car, based on the fuel consumptioncalculated in question 2? Assume that the combustion of gasoline releases 2.3 kilograms of CO2 per liter(19 pounds per gallon).arrow_forwardRefer to Table 3.3 in the lecture and take the data for water and the five other liquids listed in the table. Plot the data in a graph with the thermal conductivity on the vertical axis and the temperature range from 0°C to 100°C on the horizontal axis. Use colors or different dashed lines to make the graph presentable and understandable. The graph should look like the graph in Figure 3.1 in the lecture. Provide a short explanation of the graph in three to five sentences.arrow_forwardThe blood pressure in your heart is 1.52 x104 Pa. If an artery in the brain is 0.421 m above the heart, what is the pressure in the artery?arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





