
Concept explainers
(a)
Interpretation:
The product obtained from the reaction of cyclohexene with one equivalent of NBS in
Concept introduction:
NBS stand for
Answer to Problem 17.6P
The product obtained from the reaction of cyclohexene with one equivalent of NBS in
Explanation of Solution
It is known that the
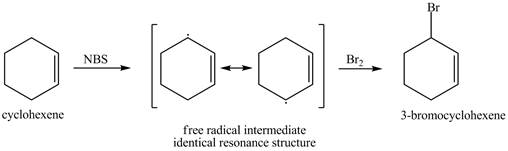
Figure 1
The product obtained from the reaction of cyclohexene with one equivalent of NBS in
(b)
Interpretation:
The products obtained from the reaction of
Concept introduction:
NBS stand for the
Answer to Problem 17.6P
The product obtained from the reaction of
Explanation of Solution
It is known that the
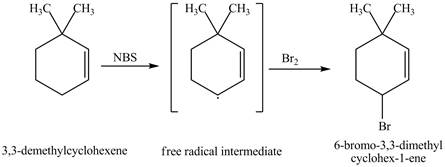
Figure 2
The product obtained from the reaction of
(c)
Interpretation:
The products obtained from the reaction of
Concept introduction:
NBS stand for the
(c)
Answer to Problem 17.6P
The products obtained from the reaction of
Explanation of Solution
It is known that the
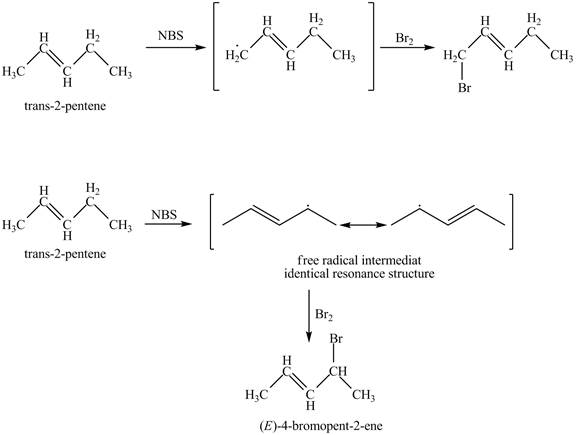
Figure 3
The products obtained from the reaction of
(d)
Interpretation:
The product obtained from the reaction of
Concept introduction:
NBS stand for the
Answer to Problem 17.6P
The product obtained from the reaction of
Explanation of Solution
It is known that the
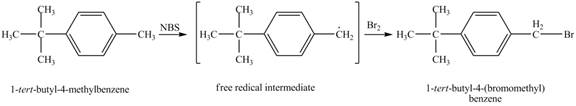
Figure 4
The product obtained from the reaction of
(e)
Interpretation:
The product obtained from the reaction of
Concept introduction:
NBS stand for the
Answer to Problem 17.6P
The product obtained from the reaction of
Explanation of Solution
It is known that the
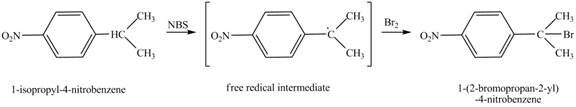
Figure 5
The product obtained from the reaction of
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- Using your reaction roadmap as a guide, show how to convert cyclohexanol into racemic trans-1,2-cyclohexanediol. (1) (2) OH aa OH (3) trans-1,2-cyclohexanediol (racemic) OH "ОН Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: (c) ethylene oxide 1 OH (a) CH3ONa+, CH, OH (d) H₂SO4, H₂O (b) OsO4, H₂O2 ||Ⓡ (9) H₂SO4 (anhydrou$)) Na, NH3 (1) (k) NBS, heat (e) m-chloroperoxybenzoic acid (mCPBA) (f) pyridinium chlorochromatę) PBr3 (PCC) (h) NaOH (N-bromosuccinimide)arrow_forwardPredict the products from reaction of 2-hexyne with the following reagents:(a) 2 equiv Br2(b) 1 equiv HBr(c) Excess HBr(d) Li in NH3(e) H2O, H2SO4, HgSO4arrow_forwardWhat product(s) would you expect from dehydration of the following alcohols with POCI3 in pyridine? Indicate the major product in each case. Он (a) CH₂CH₂CHCHCH3 I. CH3 (b) (c) .OH H CH3 H OH CH3 H3C OH II (d) CH3CHCCH₂CH3 CH3 OH (e) CH3CH₂CH₂CCH3 CH3arrow_forward
- What carbonyl compounds might you start with to prepare the follow- ing compounds by Grignard reaction? List all possibilities. (a) 2-Methyl-2-propanol (c) 3-Phenyl-3-pentanol (e) H₂C CH₂CH₂OH (b) 1-Ethylcyclohexanol (d) 2-Phenyl-2-pentanol (f) OH I -CH₂CCH3 CH3arrow_forwardDraw the structure of the organic product formed when 2,3-butanedione reacts with the following reagents. CH3 -C-C-CH3 | (a)H2/Ni (b)l2/NAOH 3Darrow_forwardWhat product would you expect from the reaction of 1 bromopropane with each Of the following? Nd also give Mechanism each of them following (a)NaNH2 (b)KOC(CH3)3 (C) Nal (d)NaCN (e)NaC=CH (f)Mg ,then H20arrow_forward
- The following sequence of steps (R)- 2-octanol to (S)- 2-octanol. Propose structural formulas for intermediates A y B, specify the absolute configuration of each, and account for the inversion of configuration in this sequencearrow_forwardHow would you prepare the following compounds from 2-phenylethanol? More than one step may be required. (a) Styrene (PHCH=CH2) (b) Phenylacetaldehyde (PHCH2CHO) (c) Phenylacetic acid (PHCH2COOH) (d) Benzoic acid (e) Ethylbenzene (f) Benzaldehyde (g) 1 -Phenylethanol (h) l-Bromo-2-phenylethanearrow_forwardPredict the products formed when CH3CH2¬C‚C≠- Na+ reacts with the following compounds.(a) ethyl bromide (b) tert-butyl bromide(c) formaldehyde (d) cyclohexanone(e) CH3CH2CH2CHOarrow_forward
- Using your reaction roadmap as a guide, show how to convert 1-butanol and ethanol into racemic 2-ethoxy-1-butanol. You must use 1-butanol and ethanol as the source of all carbon atoms in the ether product. (2) OH (1) i 2-ethoxy-1-butanol (racemic) (c) ethylene oxide a OH (a) (CH3)3 CO K+, THF (d) H₂SO4, H₂O (b) OsO4, H₂O2 OH 1 Suggest reagents and experimental conditions for each step in this synthesis. From the choices provided, suggest appropriate reagents for each step. More than one reagent may be necessary. Use the minimum number of steps possible. Enter your answer as a letter, or a series of letters, in the order necessary to bring about the steps shown. Reagents: Br OH (e) m-chloroperoxybenzoic acid (mCPBA) (f) pyridinium chlorochromate (PCC) + || (9) H₂SO4 (anhydrous) (j) Na, NH3 (1) (h) NaOH (k) NBS, heat (i) PBr3 (N-bromosuccinimide)arrow_forwardWHAT PRODUCTS WOULD YOU OBTAIN FROM REACΤION OF 1-MEΤHYLCYCLOΗEXANOL WITΗ ΤHE FOLLOWING REAGENTS? (а) НBr (b) Н>SO4 (c) CrO3 (d) Na (e) Product of part (d), then CH3Iarrow_forwardShow reagents to convert bromocyclopentane to each of the following compounds. Br OH OH (c) ОН (f) C=CH (b) Br OH (a) (d) (i) OMe OH OH "OMe (k) HO H `H.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

