
Introduction to General, Organic and Biochemistry
11th Edition
ISBN: 9781285869759
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 19.1, Problem 19.1P
Interpretation Introduction
(a)
Interpretation:
The structural formula of given amide should be drawn.
Concept Introduction:
Amides are organic compounds with −CONH2 as
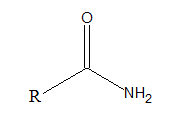
Interpretation Introduction
(b)
Interpretation:
The structural formula of given amides should be drawn.
Concept Introduction:
Amides are organic compounds with −CONH2 as functional group. The general formula of amide is.
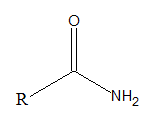
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
QUESTION 2
What is the IUPAC name of the following compound?
CH3 – CH2 – CH2 – NH-CH3
○ N-methyl-1-propanamine
○ N-propyl-1-methanamine
ethylmethylamine
diethylamine
QUESTION 3
Classify the following amine:
CH3 – CH2 – CH2 – NH – CH3
primary
seconday
O tertiary
quarternary
Is the following compound a primary, secondary, tertiary, or quaternary
amine?
H3C
N
|
CH3
A) Primary amine
B) Secondary amine
C) Tertiary amine
D) Quaternary amine
CH3
+
Question 20
The organic products from the basic hydrolysis of an amide are
O a) an amine salt and a carboxylic acid
Ob) an amide and an amide salt
O c) an amide and a carboxylic acid
Od) an amine and a carboxylic acid salt
Chapter 19 Solutions
Introduction to General, Organic and Biochemistry
Ch. 19.1 - Prob. 19.1PCh. 19.4 - Problem 19-2 Complete the equation for each...Ch. 19.4 - Prob. 19.3PCh. 19 - Prob. 19.4PCh. 19 - Write the IUPAC name for each compound.Ch. 19 - Prob. 19.6PCh. 19 - Prob. 19.7PCh. 19 - Prob. 19.8PCh. 19 - Prob. 19.9PCh. 19 - 0 Complete the equations for these reactions.
Ch. 19 - Prob. 19.11PCh. 19 - Prob. 19.12PCh. 19 - Prob. 19.13PCh. 19 - Prob. 19.14PCh. 19 - Prob. 19.15PCh. 19 - 6 Why are Dacron and Mylar referred to as...Ch. 19 - 7 What type of structural feature do the...Ch. 19 - Prob. 19.18PCh. 19 - Prob. 19.19PCh. 19 - 0 Show how triphosphoric acid can form from three...Ch. 19 - 1 Write an equation for the hydrolysis of...Ch. 19 - 2 (Chemical Connections 19A) Locate the ester...Ch. 19 - Prob. 19.23PCh. 19 - Prob. 19.24PCh. 19 - Prob. 19.25PCh. 19 - Prob. 19.26PCh. 19 - Prob. 19.27PCh. 19 - 8 (Chemical Connections 19C) Once it has been...Ch. 19 - Prob. 19.29PCh. 19 - Prob. 19.30PCh. 19 - Prob. 19.31PCh. 19 - Prob. 19.32PCh. 19 - Prob. 19.33PCh. 19 - 4 (Chemical Connections 19F) Why do Lactomer...Ch. 19 - Prob. 19.35PCh. 19 - Prob. 19.36PCh. 19 - Prob. 19.37PCh. 19 - 8 In Chapter 22, we will discuss a class of...Ch. 19 - Prob. 19.39PCh. 19 - Prob. 19.40PCh. 19 - Prob. 19.41PCh. 19 - Prob. 19.42PCh. 19 - Prob. 19.43PCh. 19 - Prob. 19.44PCh. 19 - Prob. 19.45PCh. 19 - Prob. 19.46PCh. 19 - Prob. 19.47PCh. 19 - Prob. 19.48PCh. 19 - Prob. 19.49P
Knowledge Booster
Similar questions
- NH Which of the following is the IUPAC name of the compound? Select the correct response: 1-Phenyl-N-methylethan-2-amine N-methyl-2-phenylethan-1-amine 2-Phenyl-N-methylethan-1-amine N-methyl-1-phenylethan-2-amine N-ethyl-N-methyl-1-benzenearrow_forwardWhat is the IUPAC name for the following compound? CI NH2 Multiple Choice O (E,R)-4-chloro-3-penten-2-amine (E,S)-4-chloro-3-penten-2-amine (Z,R)-4-chloro-3-penten-2-amine (Z,S)-4-chloro-3-penten-2-aminearrow_forwardGive each of the following amines an IUPAC name. (a) (b) H₂N- H3C- -N CH3 H₂ H₂ -CH3 H -C CH3 -CH3 (c) (d) Submit Answer H3C- _H₂____H₂___H__ Н NH₂ Retry Entire Group H₂ -CH3arrow_forward
- Give the IUPAC or common name of the amine formed in the reaction below between an amine salt and a base? (CH3)2NH2+Cl−+NaOH→ ?+NaCl+H2O(CH3)2NH2+Cl−+NaOH→ ?+NaCl+H2Oarrow_forwardI got two; 1 amine and nitro, Missing one more. pls help Which of the following functional groups are present in 3-nitroaniline? Group of answer choices ortho-disubstituted phenyl 1° amine nitro alkyl amide 3° amine alcohol hydroxyl 2° amine alkene ketone aldehyde ether ester meta-disubstituted phenyl para-disubstituted phenylarrow_forwardQuestion 2 of 13 Phenols are more acidic than alcohol due to Select the correct response: resonance structures positive charge polarity negative charge cannot be determinedarrow_forward
- A tertiary amine is more basic compared to a secondary amine. (A) True B Falsearrow_forwardWhat is the name of this molecule? (look at the picture to be able to answer) a) N, N, N-ethylmethylpentylamine b) 2-amine-2-methylheptane c) N-ethyl-N-methylpentanamine d) N-ethyl-N-methylpentamide e) ethylmethylpentylamine f) 1-methylethylpropylamide g) None of the answers are correct.arrow_forwardQuestion 4 An amide cannot be synthesized from the following combinations: O Carboxylic acid + ammonia Carboxylic ester + a primary amine Dehydration of nitriles O Acyl chloride + ammoniaarrow_forward
- Course Home f11 P AAI ? f12 { ins 76°F Mostly cloudy ^ 0 0 6 ENG prt sc pause delete ← backspace enter home T shift lock 7 home ↑ A 4 end 1 9:10 PM 4/14/2023 -00 8 pg up 5 9 pg 2arrow_forwardQuestion 28 Saponification product of butylpropanoate is: A Propanol and Sodium butanoate B Butanol and Propanoic acid C Butanol and sodium propanoate Butanoic acid and Propanoic acidarrow_forwardCh 11 Home de Dates > Ch 11 Homework O Resources Draw the products formed when the given amides are treated with aqueous potassium hydroxide. Reaction A Select Draw Rings More Erase OH КОН N. about us careers privacy policy terms of use contact us help (? SAMSUNG logitech O PO Print Screen Serol Lock Pause Break F12 F11 F9 F10 F8 F6 F7 F5 Home Page Up Insert 14arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning