
To review:
The chemical structures that are involved in the ionization of acetic acid in water.
Introduction:
The water is a polar and universal solvent. Most of the molecules having ionic bonds dissolve in water by making bonds with it. The compound dissociates into the individual ions making the ionic bond weaker than the solid form of the compound.
Explanation of Solution
The acetoacetic acid dissociates into the acetoacetate
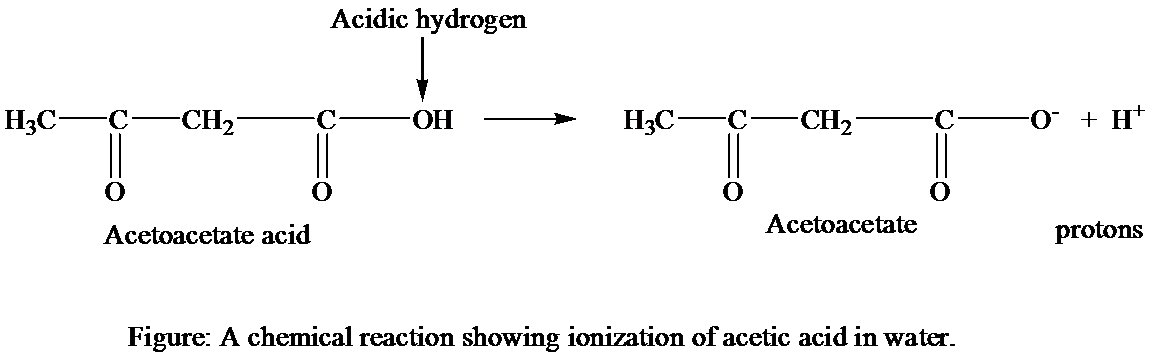
Thus, it can be concluded that the acetoacetate and hydrogen ions are chemical structures that are involved in the ionization of acetoacetic acid in water. The carboxyl group of acetoacetate infers the negative charge on the molecule and hydrogen ion gets dissociated from acetoacetic acid with its positive charge.
Want to see more full solutions like this?
Chapter 2 Solutions
Life: The Science of Biology
- Identify the conjugate acid-base pairs in the following reactions: HNO2(aq) + H2O(l) → NO2 – (aq) + H3O+(aq) _______ ______ _________ ________ CH3NH2 + H2O(l) → CH3NH3+ + OH – _______ ________ ________ _________arrow_forwardThe following compounds are to be synthesized through a mixed aldol conduction reaction. Draw two chemical structures of aldehyde or ketone.arrow_forwardIn solution, glucose exists predominantly in the cyclic hemiacetal form, which does not contain an aldehyde group. How is it possible for mild oxidizing agents to oxidize glucose?arrow_forward
- Write the structural formula for the product of the reaction.arrow_forwardFumaric acid is a metabolic intermediate that has the systematic name trans-2-butenedioic acid. Draw its structure.arrow_forwardUsing the picture provided, match the correct name of each of the functional groups highlighted in blue.arrow_forward
- Identify the major and minor product(s) of the following reaction:arrow_forwardWrite a stepwise mechanism for the following reaction.arrow_forwardWrite the complete reaction for the isomerization of citrate. Name the enzyme that catalyses these reaction andcprovide the formulas of the reactants and the product of these reaction ?arrow_forward
- Give 5 examples of molybdenum complexes. State their physical and chemical properties and their importance or applications. Additional question, what are the oxidation numbers of molybdenumarrow_forwardPlease draw the major product(s) of the following reaction and show the mechanism. Is there a reason why one stereochemistry is preferred over the other for this reaction?arrow_forwardDehalogenase enzymes catalyze the clevage of C-X bonds. One such dehalogenase catalyzes the following reaction. An active site aspartate is thought to carry out the initial nucleophilic attack that expels the chloride. Select the most likely intermediate in the reaction. (Note the stereochemistry. Refer to P11.19 in the textbook if needed.) O O A C D OE -CH₂ H H₂C™ CO₂ H B CO₂ CI N H₂O Cr CO₂ H CO₂ нотум CH3 D CO₂ five N E CH₂arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





