
Concept explainers
Draw a “formula” for each of the following molecules using circular symbols of your choice to represent atoms:
a. A diatomic molecule of an element
b. A diatomic molecule of a compound
c. A triatomic molecule of an element
d. A molecule of a compound containing one atom of one element and four atoms of another element
(a)
Interpretation:
The formula for the diatomic molecule of an element by using circular symbols to represent atoms is to be drawn.
Concept introduction:
A molecular formula represents the number of atoms of each element present in a molecule of a compound.
The number of atoms present in molecule is determined by the subscript written below the normal line in the molecular formula.
Answer to Problem 2.1E
The formula for the diatomic molecule of an element by using circular symbols to represent atoms is shown below.
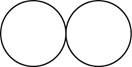
Explanation of Solution
It is given that the molecule of an element is a diatomic which means that the compound consists of two atoms with similar identity. This can be explained with the help of one example. Considering a diatomic molecule of an element that is chlorine gas. The chemical formula of chlorine gas is
Therefore, the formula for the diatomic molecule of an element by using circular symbols to represent atoms is shown below.

Figure 1
In the given figure, white circles represent the chlorine atoms.
The formula for the diatomic molecule of an element by using circular symbols to represent atoms is shown in figure 1.
(b)
Interpretation:
The formula for the diatomic molecule of a compound by using circular symbols to represent atoms is to be drawn.
Concept introduction:
A molecular formula represents the number of atoms of each element present in a molecule of a compound.
The number of atoms present in molecule is determined by the subscript written below the normal line in the molecular formula.
Answer to Problem 2.1E
The formula for the diatomic molecule of a compound by using circular symbols to represent atoms is shown below.
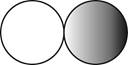
Explanation of Solution
It is given that the molecule of a compound is diatomic which means that the compound consists of two different atoms. This can be explained with the help of one example. Considering an example that is hydrogen fluoride. The chemical formula of hydrogen fluoride is
Therefore, the formula for the diatomic molecule of a compound by using circular symbols to represent atoms is shown below.
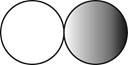
Figure 2
In the given figure, grey circle represent the fluorine atom, whereas white circle represent the hydrogen atoms.
The formula for the diatomic molecule of a compound by using circular symbols to represent atoms is shown in figure 2.
(c)
Interpretation:
The formula for the triatomic molecule of an element by using circular symbols to represent atoms is to be drawn.
Concept introduction:
A molecular formula represents the number of atoms of each element present in a molecule of a compound.
The number of atoms present in molecule is determined by the subscript written below the normal line in the molecular formula.
Answer to Problem 2.1E
The formula for the triatomic molecule of an element by using circular symbols to represent atoms is shown below.
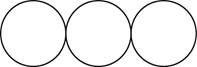
Explanation of Solution
It is given that the molecule of an element is a triatomic which means that the compound consists of three atoms with similar identity. This can be explained with the help of one example. Considering an example that is ozone. The chemical formula of ozone is
Therefore, the formula for the triatomic molecule of an element by using circular symbols to represent atoms is shown below.

Figure 3
In the given diagram, white circles represent oxygen atoms.
The formula for the triatomic molecule of an element by using circular symbols to represent atoms is shown in figure 3.
(d)
Interpretation:
The formula for a molecule of a compound containing one atom of one element and four atoms of another element by using circular symbols to represent atoms is to be drawn.
Concept introduction:
A molecular formula represents the number of atoms of each element present in a molecule of a compound.
The number of atoms present in molecule is determined by the subscript written below the normal line in the molecular formula.
Answer to Problem 2.1E
The formula for a molecule of a compound containing one atom of one element and four atoms of another element by using circular symbols to represent atoms is shown below.
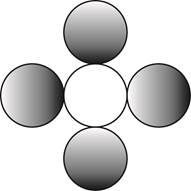
Explanation of Solution
It is given that the molecule contains one atom of one element and four atoms of another element. This can be explained with the help of one example. Considering an example that is carbon tetrachloride. The chemical formula of carbon tetrachloride is
Therefore, the formula for a molecule of a compound containing one atom of one element and four atoms of another element by using circular symbols to represent atoms is shown below.
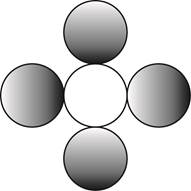
Figure 4
In the given diagram, white circle represent carbon atom, whereas grey circle represent chlorine atoms.
The formula for a molecule of a compound containing one atom of one element and four atoms of another element by using circular symbols to represent atoms is shown in figure 4.
Want to see more full solutions like this?
Chapter 2 Solutions
Chemistry for Today: General, Organic, and Biochemistry
Additional Science Textbook Solutions
General, Organic, and Biological Chemistry - 4th edition
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Introduction to Chemistry
Chemistry: Structure and Properties (2nd Edition)
- Determine the number of each type of atom in molecules represented by the following formulas: a. sulfur dioxide (SO2) b. butane (C4H10) c. chlorous acid (HClO2) d. boron trifluoride (BF3)arrow_forwardInterpreting Chemical Formulas Determine the number of each type of atom in the following chemical formulas: CCl4 Al2(SO4)3arrow_forwardb and c and naming the moleculesarrow_forward
- Part A: Pairs of ions are attracted to each other to build an ionic ________ Your answer Part B: Pairs of ions are attracted to each other to build an ionic ________ Part C: What is the formula for Sodium Chloride? Part D: What is the ratio of Sodium ions to Chloride ions in Sodium Chloride? Your answerarrow_forwardQUESTION 1 When sodium chloride dissolves in water, how do the water molecules orient around the ions? O A. Water molecules are randomly oriented around the ions. O B. The hydrogen atoms point toward both the sodium and the chloride. O C. The oxygen atoms point toward both the sodium and the chloride. O D. Around sodium the hydrogen atoms point toward the sodium, and around chloride the oxygen atoms point toward the chloride. O E. Around sodium the oxygen atoms point toward the sodium, and around chloride the hydrogen atoms point toward the chloride.arrow_forwardThe element potassium forms a _______cationanion with the charge . The symbol for this ion is , and the name is . The number of electrons in this ion is .arrow_forward
- Table 4 shows data from an experiment to determine the formulas of hydrated barium chloride. Determine the formula for the hydrate and its name.arrow_forward37. On a certain day a financial website quoted the price of gold at 1,258 per troy ounce (1troyounce=31.1g). What is the price of a single atom of gold (Z=79)?arrow_forwarda How many atoms of hydrogen are present in 2.69moles of water? b How many moles of oxygen are present in 1.391022molecules of water?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





