
Concept explainers
(a)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(a)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 1.
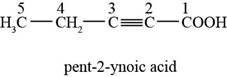
Figure 1
The structure comprises chain of five carbon atoms along with
Therefore, the
(b)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in an electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(b)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 2.
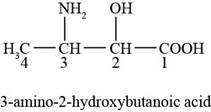
Figure 2
The structure comprises chain of four carbon atoms along with carboxylic acid group, hydroxyl group and amino group at first, second, and third carbon atoms respectively. Carboxylic acid group gets higher priority during the naming of structure because it is present at first carbon atom. According to
Therefore, the
(c)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in an electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(c)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 3.
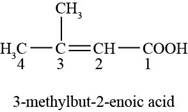
Figure 3
The structure comprises chain of four carbon atoms along with carboxylic acid group and methyl group at first and third carbon atoms respectively. The double bond is present in between the second and third carbon atoms. Carboxylic acid group gets higher priority during the naming of structure because it is present at first carbon atom. According to
Therefore, the
(d)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in an electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(d)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 4.
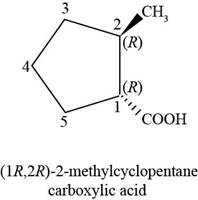
Figure 4
The structure comprises cyclic ring of five carbon atoms along with one group of carboxylic acids at first carbon atom and one methyl group at second carbon atom. The carboxylic acid group is present below the plane, whereas methyl group is present at above the plane. According to
Therefore, the
(e)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(e)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 5.
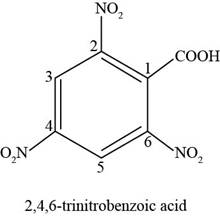
Figure 5
The structure comprises cyclic ring of six carbon atoms along with one carboxylic acid group at first position and three nitro groups at second, fourth and sixth carbon atoms. According to
Therefore, the
(f)
Interpretation:
The IUPAC name of given compound is to be stated.
Concept introduction:
Carboxylic acids are the carbon compounds that contain carboxyl group as a major functional group. These are polar compounds due to difference in electronegativity of the atoms in a compound.
The chemical structures are described by IUPAC name or common names. IUPAC names are totally different from common names because IUPAC names are applied at international level and it comprises suffix, prefix, numbers and other priority rules.
(f)
Answer to Problem 20.25SP
The
Explanation of Solution
The chemical structure of the given compound is shown in figure 6.
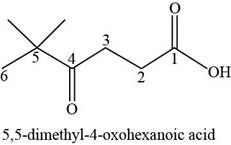
Figure 6
The structure comprises parent chain of six carbon atoms along with carboxylic acid group, carbonyl group, and two methyl groups at first, fourth and fifth carbon atoms respectively. According to
Therefore, the
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry Plus Masteringchemistry With Pearson Etext, Global Edition
- College Chem II CHE 132 Alcohols & Ethers - Naming & Reactions 1. Name the following compounds. For a-d, indicate whether the compound is 1,2, or 3. b) CH a) CH-CH-CH-CHy CH-al-CH-CHy d) OH c) CHy f) CHy-CH- CH-CHy e) CHICHS 8) CHy-CH-CHy h) CHy-CH-0-CH-CHICHY CH2 CHy-CH-CH3arrow_forwardCH3 KOC(CH 3)3 H;C- -CH3 Solvent: Ethanol HCI c-OH Solvent: Ethanol CH3 KOC(CH 3)3 Solvent: Toluene H3C- -CH3 HCI CH3 но CH3 Solvent: Toluene H3C H3C CH3 KOC(CH 3)3 .CI Solvent: Toluene -CH3 CH3arrow_forwardDetermine the products of each carboxylic acid reaction. a. CH3-CH2-CH2-CH,-C-OH + H,SO, CH, — CH,— он HO. b. CH2 heat CH2 C-OH CH2arrow_forward
- 2. Write the equations: a) CH3 -CH-CH-COOH + Ca → CH, NH, b) CH3 -CH-CH – COOH + CH₂OH- CH, NH, c) CH3 -CH-CH-COOH + Ba(OH)₂ CH, NH, d) CH3 -CH-CH-COOH + HCI → CH, NH, e) CH3 -CH-CH-COOH- CH, NH, [0] OH [H] LIAIH4 H* +tº f) CH3 -CH-CH-COOH + CH3 -CH-CH-COOH- -H₂O CH, NH, NH₂ g) CH3 -CH-CH-COOH- CH3NH₂arrow_forwardWhich alkyl halide has the highest boiling point? A. CH3BrB. CH3FC. CH3ClD. CH3larrow_forwardGive the IMPAC name for each compound. 1. COOH COOH 2. Br 3. CH3 CHz COOHarrow_forward
- Complete the following halogenation reactions for alcohols. Draw the structure of the product. Name the reactant and the product. a) Cty-CH-0H b) CH3 CH-CH-CH PBY3 c) PE 7. Name the following phenols. d) a) b) OH c) OH Br OH CH2 HO CI Br 8. Name the following ethers. b) a) CHy-0-C-CHs CHy-0-CH3 c) d) Cy-CH-0 talamc) Earrow_forwardCH,-C-CHCH, Classify each compound as an aldehyde or a ketone. Give the common name for each ketone. CH,CH,CH,-C-H CH,CH,CH,-C-CH,CH,CH, CH, CH;-C-CHCH, Name the following organic compound containing oxygen. CH, O CH, 3, | CH,CH-C-CHCH, || CH,CH,CH,-C-CH,CH, CH3 || CH,CH,CH,CH-Ĉ-H CH3 CH,CH,CH-C–Harrow_forwardOH H3C Type of Reaction: CH3 CH3 CH3 + OH Type of Reaction: + [0] НОarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
