
Concept explainers
(a)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic
(a)
Explanation of Solution
Given compounds,
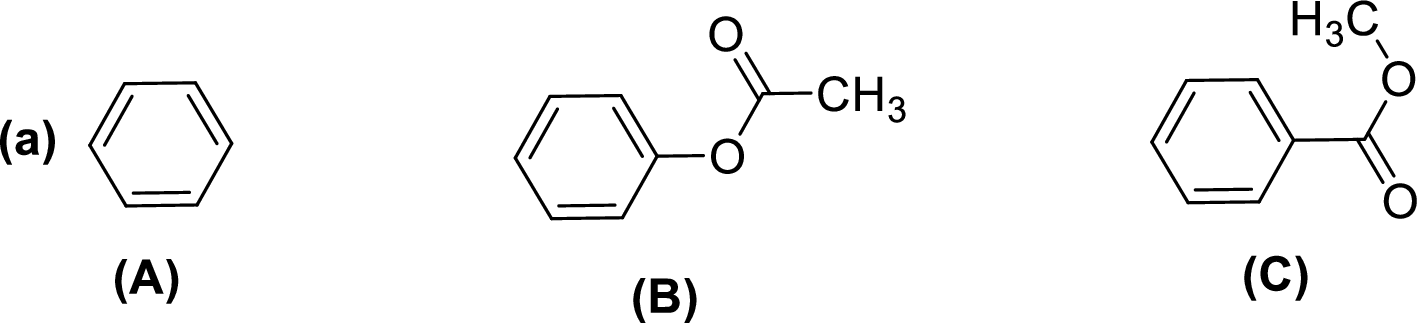
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(b)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(b)
Explanation of Solution
Given compounds,
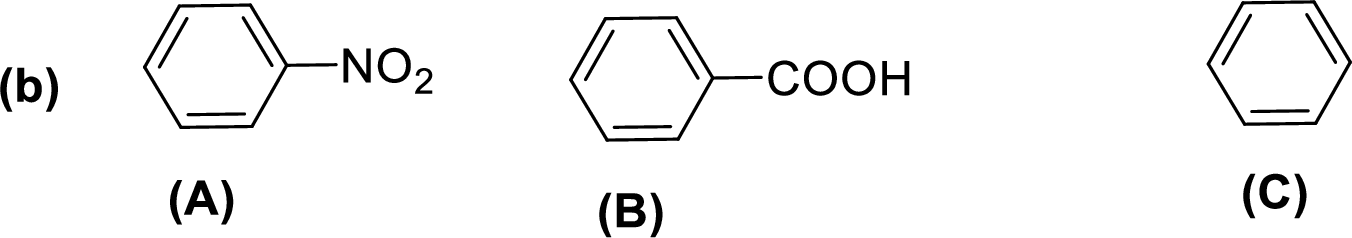
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(c)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(c)
Explanation of Solution
Given compounds,
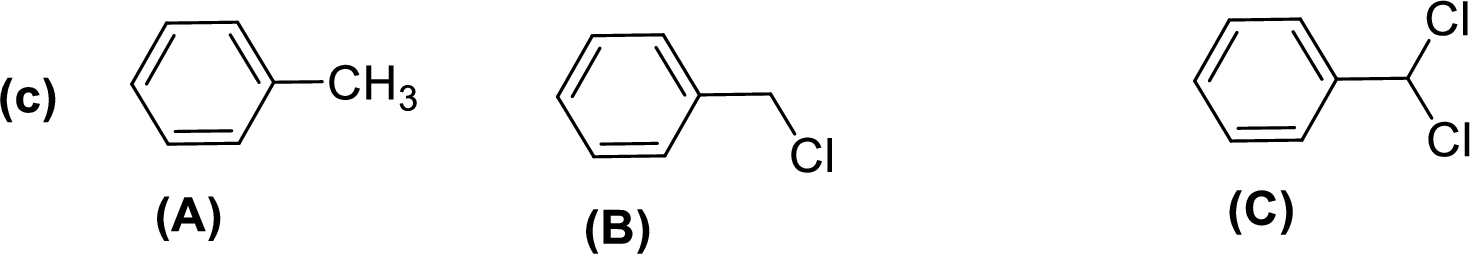
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(d)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(d)
Explanation of Solution
Given compounds,
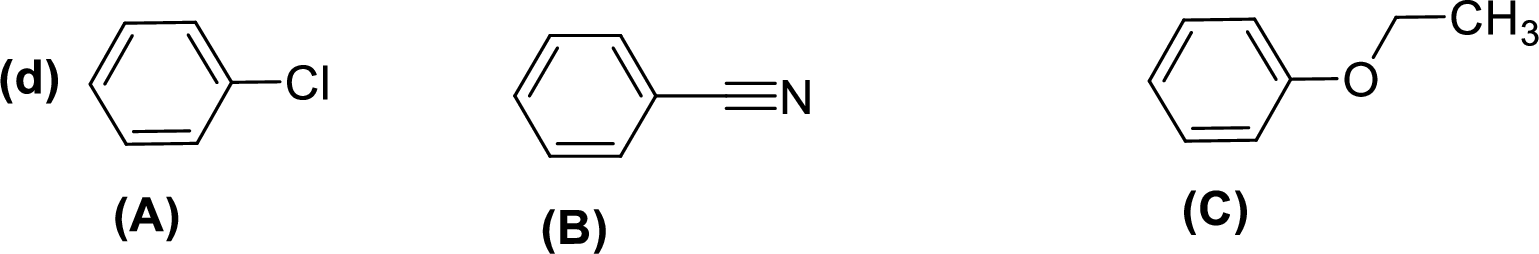
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
(e)
Interpretation:
Given compounds has to be arranged in their decreasing order of reactivity towards electrophilic aromatic substitution.
(e)
Explanation of Solution
Given compounds,
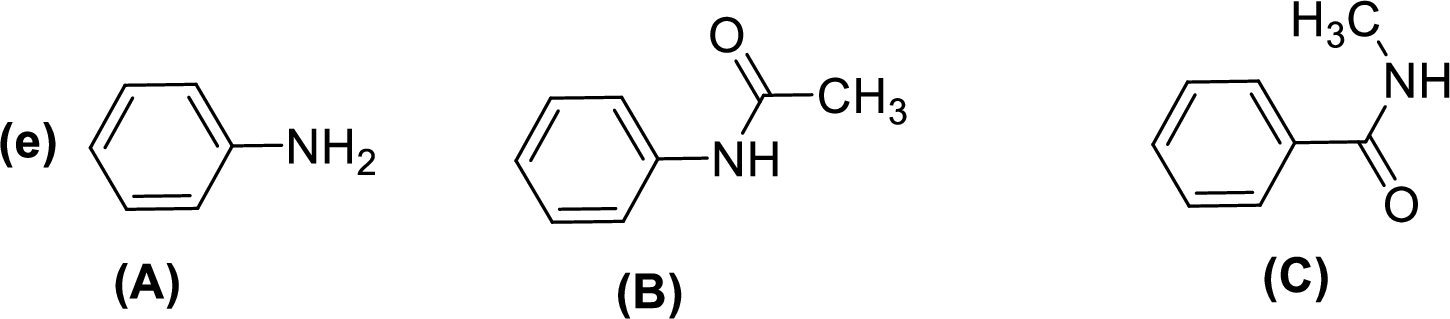
General trend of compounds reactivity in electrophilic aromatic substitution for a set of compounds having similar structure is given by,
So, electron donating substituents will increases the reactivity, while electron withdrawing substituents will decreases the reactivity.
Therefore, the decreasing order of reactivity towards electrophilic aromatic substitution of the given compounds are given below,
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- Both pyridine and pyrrole are nitrogen containing aromaticheterocyclic compounds. When treated with HCI, only pyridine forms a hydrochloride salt, whereas pyrrole is unreactive Which of the following is not a valid explanation for this observed reactivity? pyridine ругrole O The lone pair on pyridine is not part of the aromatic system. O The lone pair on pyridine can be protonated without disrupting the aromatic stability. O The lone pair on pyrrole is sp hybridized and is less prone to protonation. O Protonation of pyrrole leads to a nonaromatic cation, which is less stable O The lone pair on pyrrole is involved in making the compound aromatic and thus is less susceptible to protonation.arrow_forwardRank the following compounds in order of increasing reactivity toward nucleophilic attack.arrow_forwardRank group of compounds from most reactive to least reactive toward electrophilic aromatic substitution: :- benzene, benzoic acid, phenol, propylbenzenearrow_forward
- Rank the compounds in attached group in order of increasing acidity.arrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict themajor products of the reactions of naphthalene with the following reagents. f) fuming sulfuric acidarrow_forwardRank the attached compounds in order of increasing basicity andexplain the order you chose.arrow_forward
- Which of the following is the correct order of decreasing reactivity towards electrophilic aromatic substitution? Phenol > benzene > propylbenzene > benzoic acid Phenol > propylbenzene > benzene > benzoic acid Propylbenzene > benzene > benzoic acid > phenol Propylbenzene > benzoic acid > phenol > benzenearrow_forwardConsider carbonyl compounds A–E drawn below. (a) Rank A–E in order of increasing stability. (b) Rank A–E in order of increasing amount of hydrate formed when treated with aqueous acid. (c) Which compound is most reactive in nucleophilic addition?arrow_forwardRank the attached compounds in order of increasing basicity.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





