
Interpretation: Each amide group present in oxytocin is to be classified as
Concept introduction: The amide is identified as primary
Answer to Problem 22.1P
The classification of each amide group present in oxytocin as
Explanation of Solution
The classification of each amide group present in oxytocin as
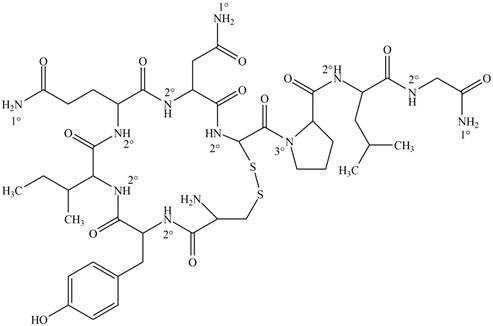
Figure 1
Amides are the organic compounds which contain
The classification of each amide group present in oxytocin as
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry
- What carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.arrow_forwardClassify the specified amides as 1º, 2º, or 3º. Part 1 Select the single best answer. Part 2 Oxytocin, sold under the trade name Pitocin, is a naturally occurring hormone used to stimulate uterine contractions and induce labor. Classify the specified amides in oxytocin as 1º, 2º, or 3º.arrow_forwardFenfluramine and phentermine are two components of fen–phen, an appetite suppressant withdrawn from the market in 1997 after it was shown to damage the heart valves in some patients. What products are formed when fenfluramine and phentermine are each treated with acetic acid (CH3CO2H)?arrow_forward
- Acetaminophen is an analgesic marketed under the brand name Tylenol, among others. Draw the amine that results from the base hydrolysis of acetaminophen.arrow_forwardWhat ammonium salt is formed when each amine is treated with HCl? Draw the structure of the resulting salt.arrow_forwardGive a systematic name for each amide ?arrow_forward
- Explain why many amines with useful medicinal properties are sold as their ammonium saltsarrow_forwardIndicate whether the following statement is true or false. Aliphatic amines are more basic than ammonia, whereas aromatic amines are less basic than ammonia.RightFalsearrow_forwardWhat products are formed when each amide is hydrolyzed with water and HCl?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,



