
Concept explainers
(a)
Interpretation:
The function of the sodium hydride has to be determined in the 1st step along with the
Concept introduction:
Aromaticity:
Aromaticity is a property of cyclic, planar structures with a ring of resonance bonds that gives increased stability compared to the other geometric or connective arrangements with the same set of atoms. Aromatic molecules are very stable and do not easily break up to take part in reactions.
There are some rules for a compound to be aromatic i.e. they have to be planar, cyclic and must contain
(a)
Explanation of Solution
In the given question the 1st step is,
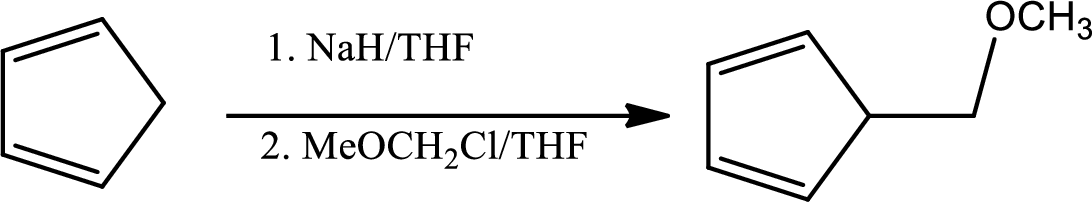
The mechanism as follows,
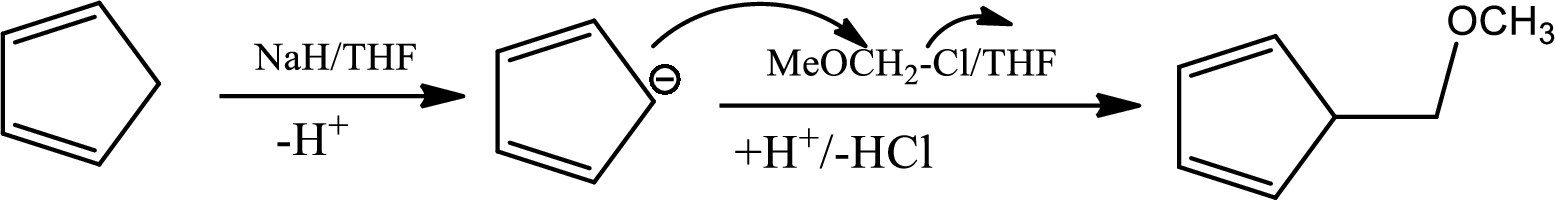
Here
The
(b)
Interpretation:
The reaction pathway by which B is converted to C that has to be identified.
Concept introduction:
Diels Alder reaction:
The Diels-Alder reaction is a
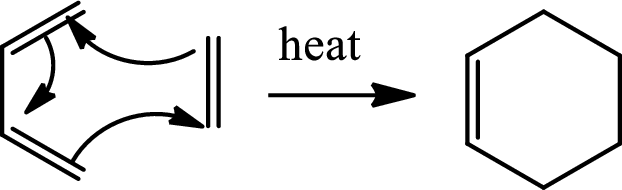
(b)
Explanation of Solution
According to the question,
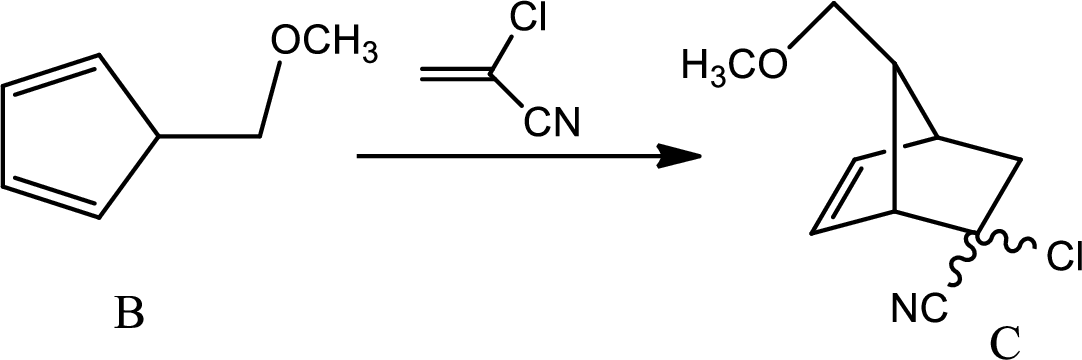
Here B is the diene and the dienophile consists of electron withdrawing groups.
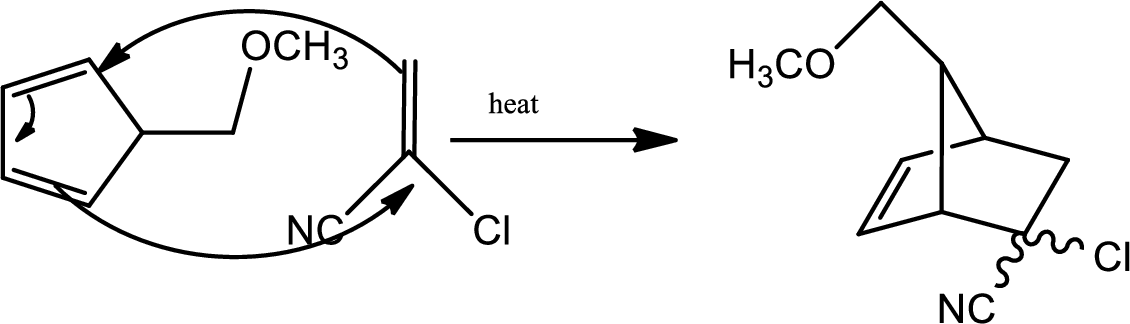
Thus via Diels Alder reaction the product is obtained.
(c)
Interpretation:
The role of the carbon dioxide added to the reaction mixture in step 2 of the conversion of (E) to (F) has to be determined.
Concept introduction:
Lactone formation:
Lactones are cyclic carboxylic esters that are formed by intramolecular esterification of the corresponding hydroxycarboxylic acids which takes place spontaneously when th ering that is formed is five or six membered. The reaction pathway is as follows,
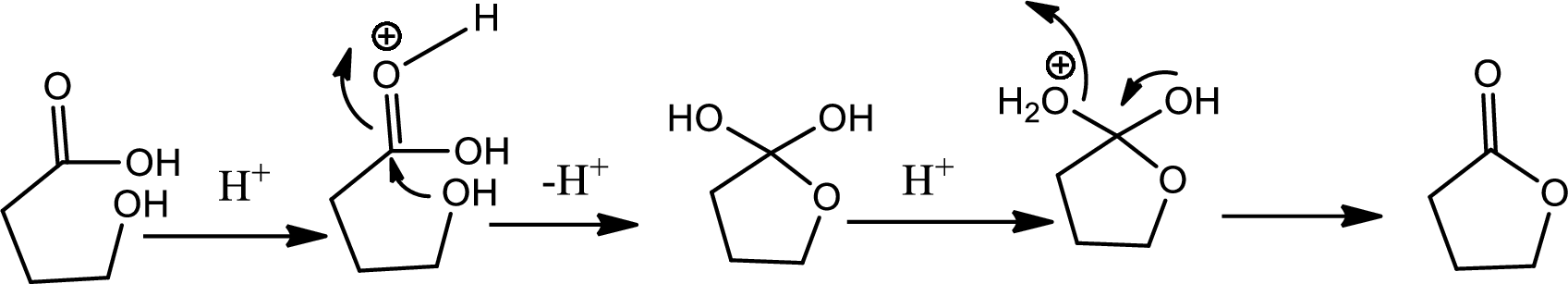
(c)
Explanation of Solution
The reaction given is,
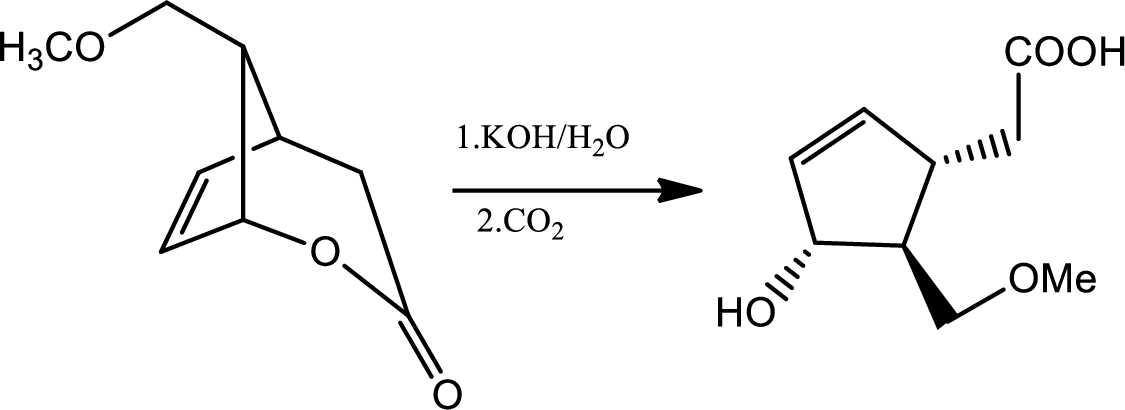
In this reaction in the presence of base lactone is hydrolised to give back acid and alcohol. Now if the amount of base is more in medium then again the backward reaction will be favoured and lactone will be formed again. So the reactant will be formed again. Hence to maintain the
Carbon dioxide reacts with water to give mild amount of carbonic acid that dissociates in water to a little extent to give less amount of proton which is sufficient to balance the
(d)
Interpretation:
A reaction pathway has to suggest for the conversion of (H) to (I) via radical mechanism.
Concept introduction:
Radical reaction:
A free radical reaction is a chemical reaction involving free radicals. Radical reaction contain three steps that are respectively chain initiation, chain propagation and chain termination.
(d)
Explanation of Solution
The reaction given,
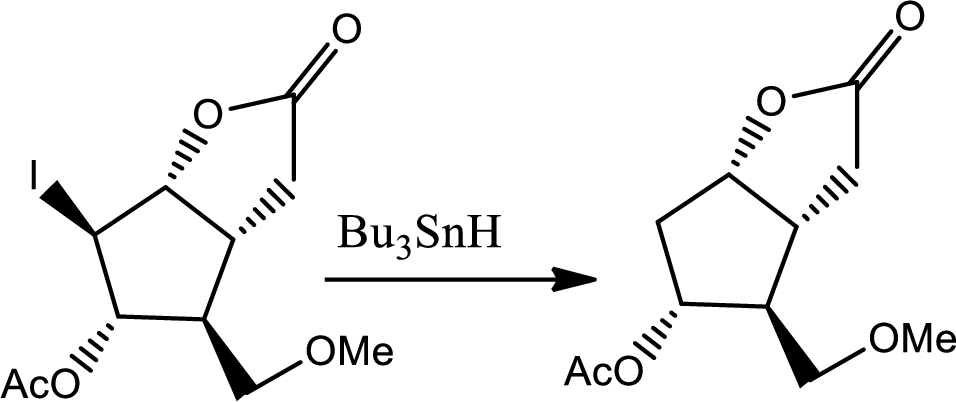
According to the question, the reaction proceeds via radical pathway and the 1st step involves a radical initiator to form
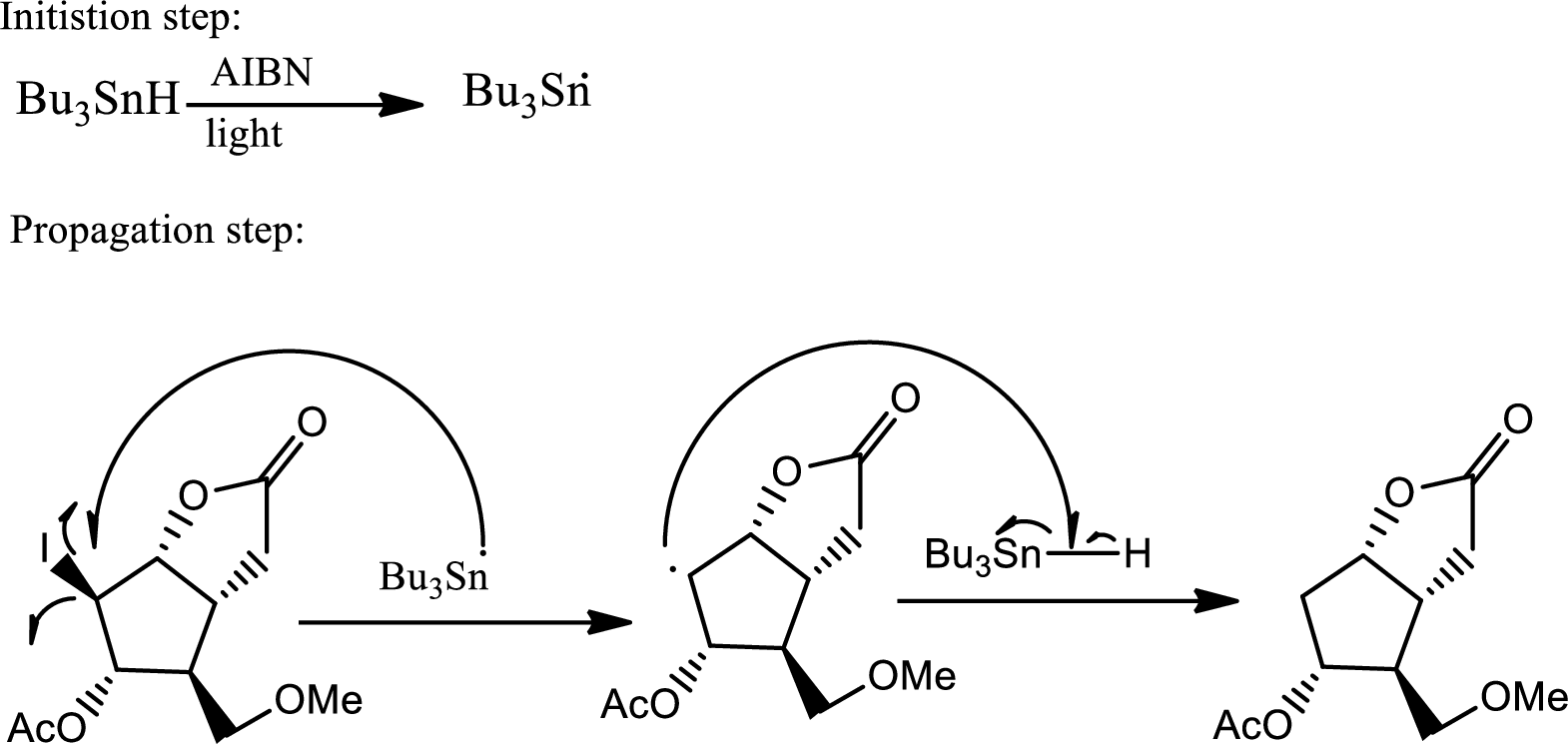
As a radical initiator AIBN is used. The reaction undergoes via two steps which are initiation and propagation respectively. Initiation step involves a radical initiator to form
(e)
Interpretation:
The steps in which the chiral centers of Corey lactone can be determined has to be shown with the mechanism.
Concept introduction:
Chiral centre:
Chiral centre is defined as an atom bonded to four different chemical species. It is a stereo centre that holds the atom in such way that the structure may not be superimposable to its mirror image. They give optical isomerism.
(e)
Explanation of Solution
In the question it is given that the Corey lactone has four chiral centers which are pointed below,
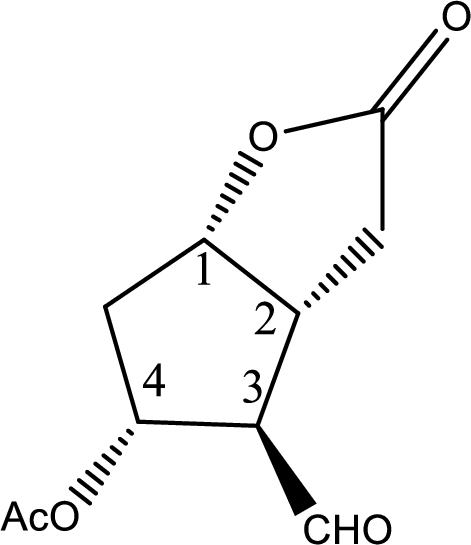
Now the steps where the chiral centers can be distinguished are given below,
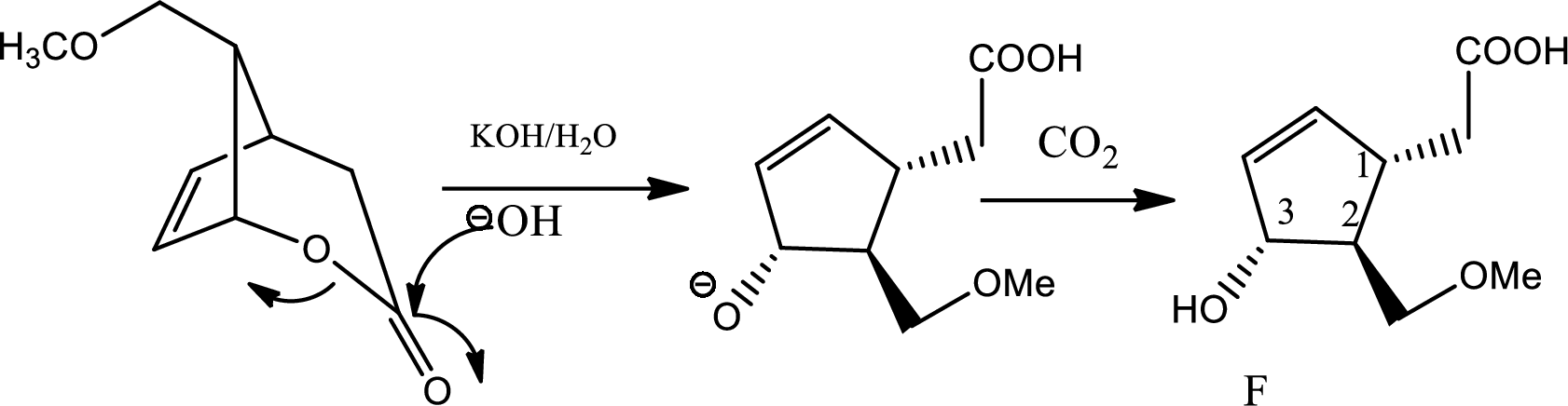
In this step due to hydrolisation of lactone, ring opening occurs along with the formation of acid and alcohol.
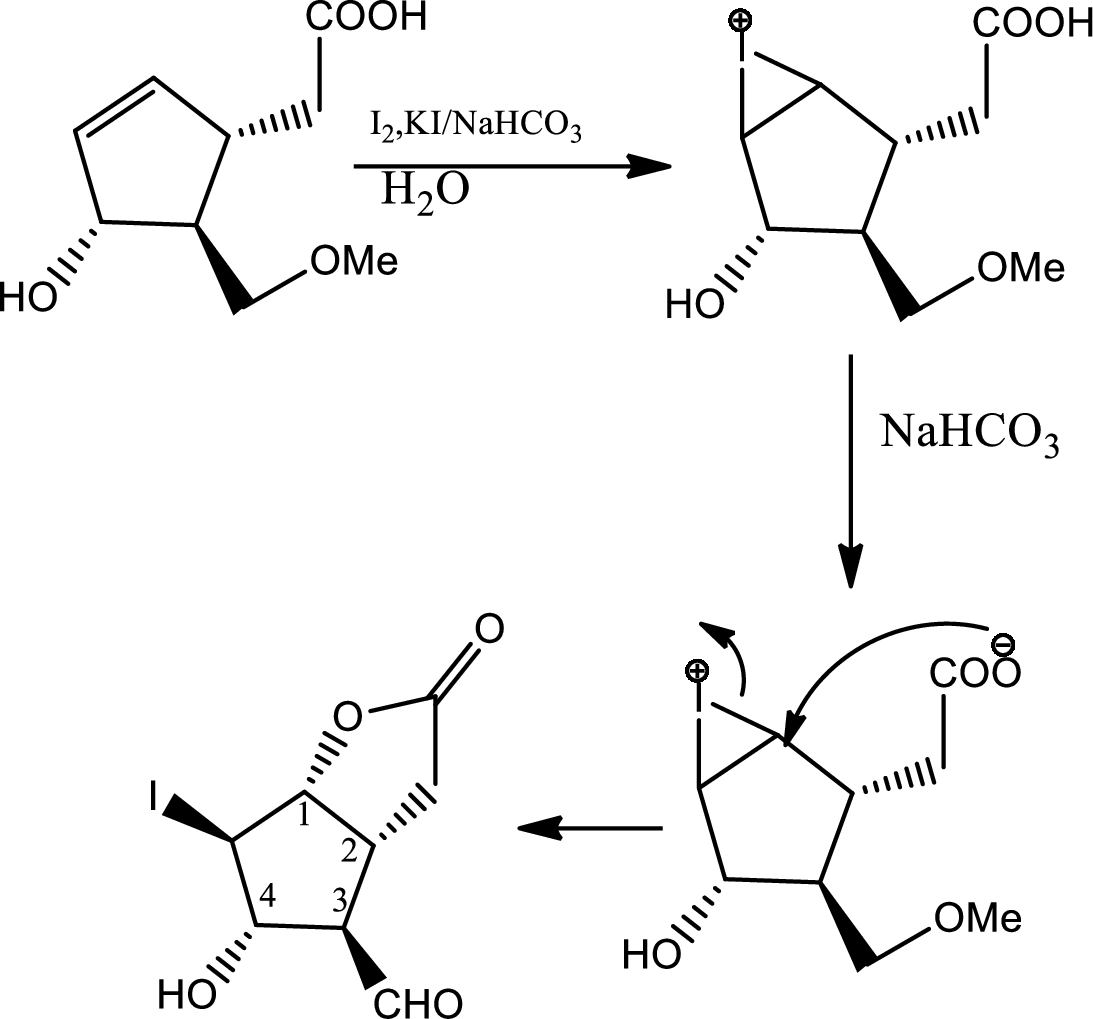
In this step normal
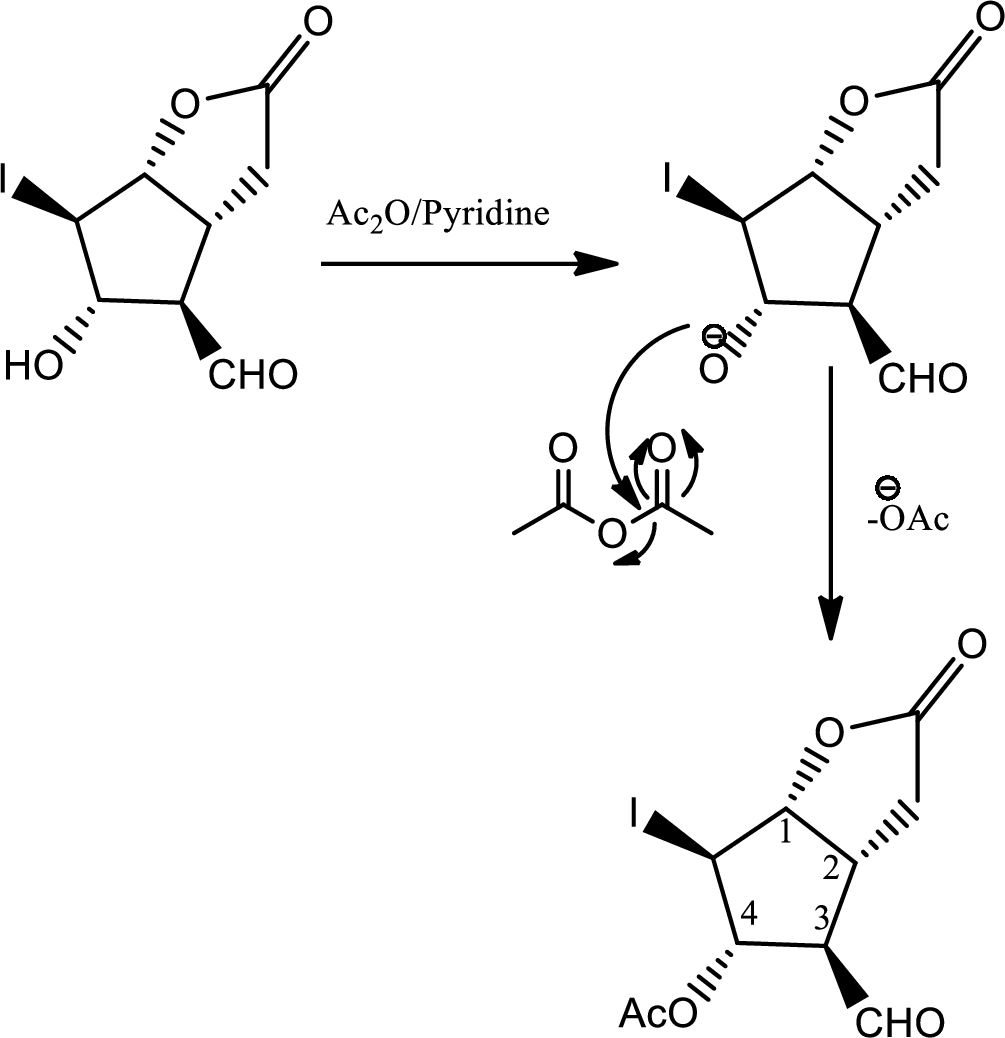
In this step normal acid base reaction occurs followed by normal
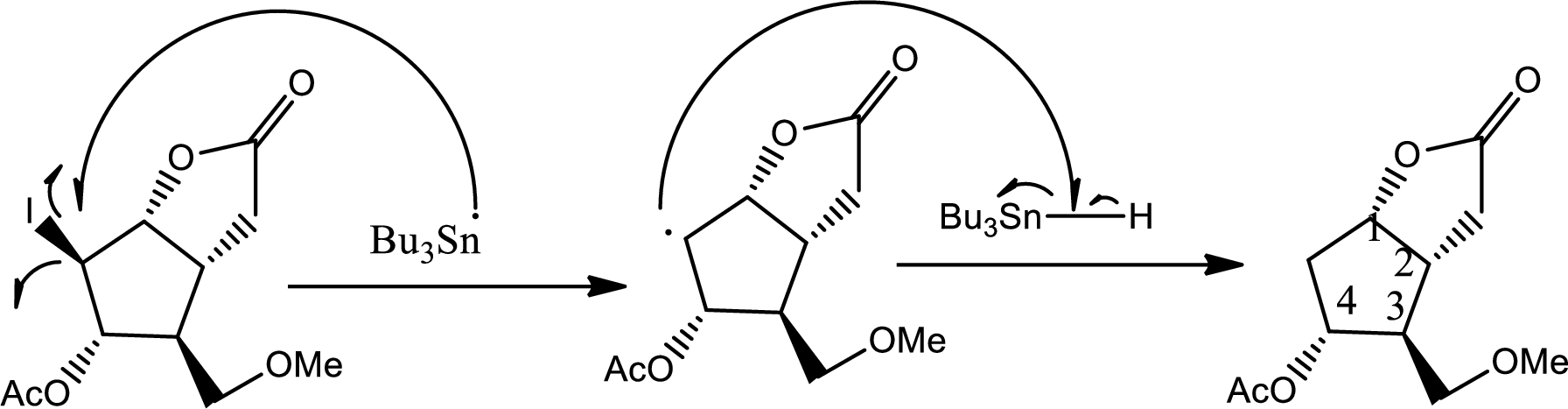
Via radical mechanism this step occurs to break the carbon-halogen bond.
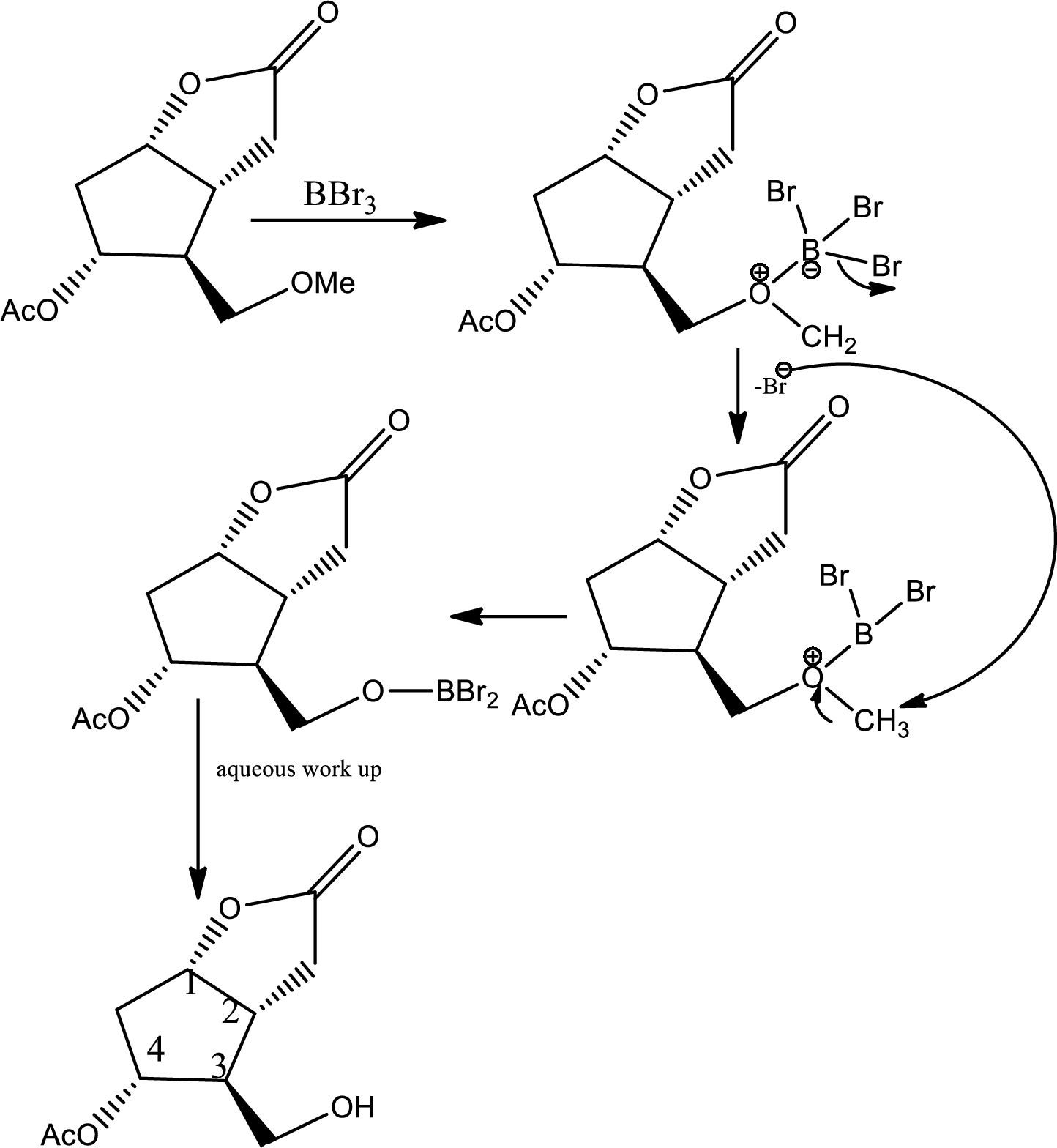
Here the ether is converted to alcohol.
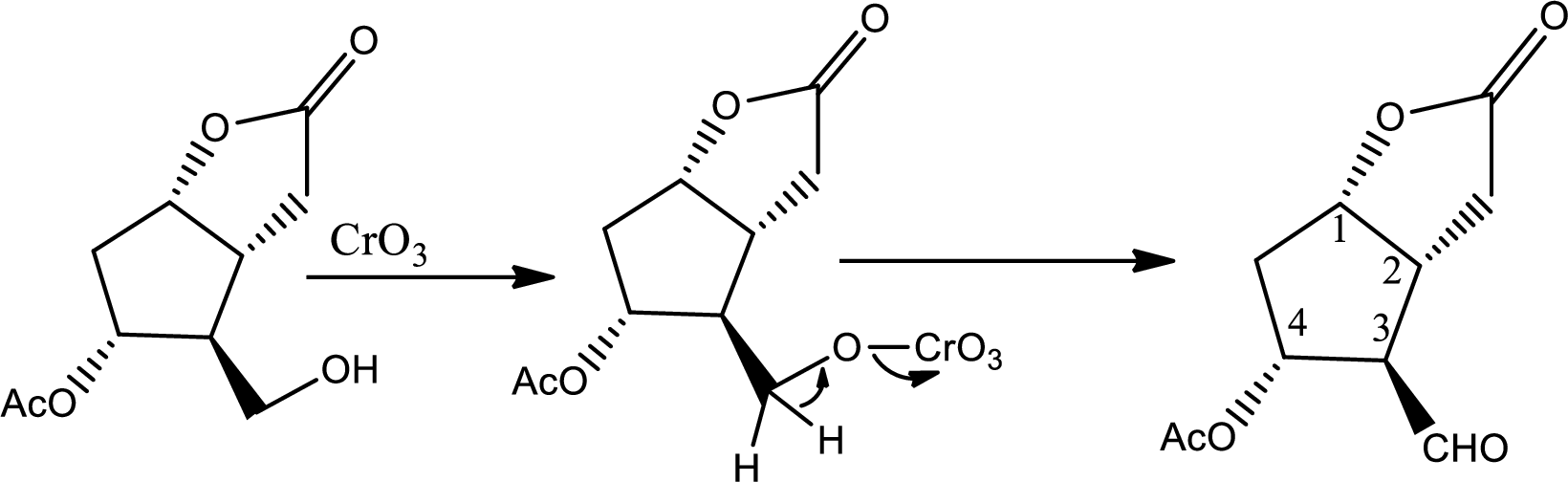
Here alcohol is oxidized to
In all these steps the chiral centres can be distinguished.
(f)
Interpretation:
The definition of resolution has to be given along with the rationale for using a chiral, enantiomerically pure
Concept introduction:
Resolution:
Chiral resolution in stereochemistry is a process for the seperation of racemic compounds into their enantiomers. Now racemic mixture is the mixture that has equal amounts of left and right handed enantiomers of the chiral molecule.
(f)
Explanation of Solution
According to the question the compound F formed is resolved by (+)-ephedrine. The structures of compound (F) and (-)-ephedrine are given below,
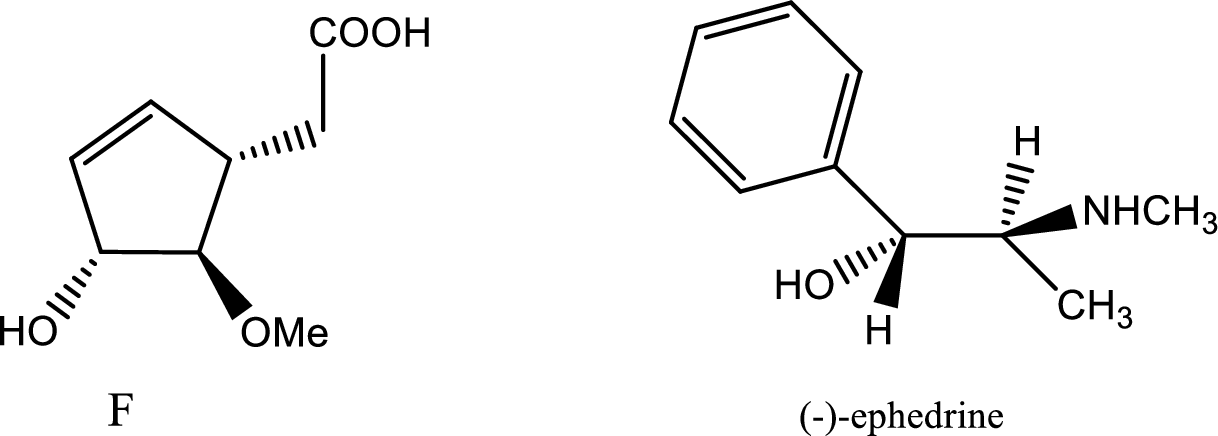
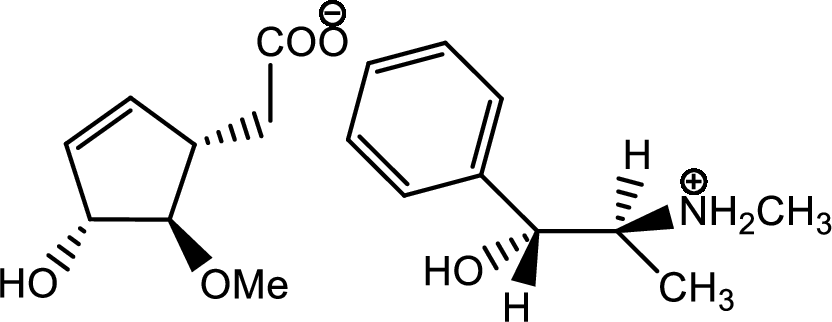
The process of racemisation follows acid base reaction between compound F and (+)-ephedrine. The two enantiomers can be converted to two diastereomeric salts those are [(+)(+)] and [(-)(+)]. Now being diastereomers they can be easily separated as diastereomers have different physical properties. Thus racemisation process works to separate two enantiomers.
One of the salts is shown below,
(g)
Interpretation:
The mechanism for the reaction pathway from D to E that undergoes via Baeyer-Villiger oxidation has to be shown.
Concept introduction:
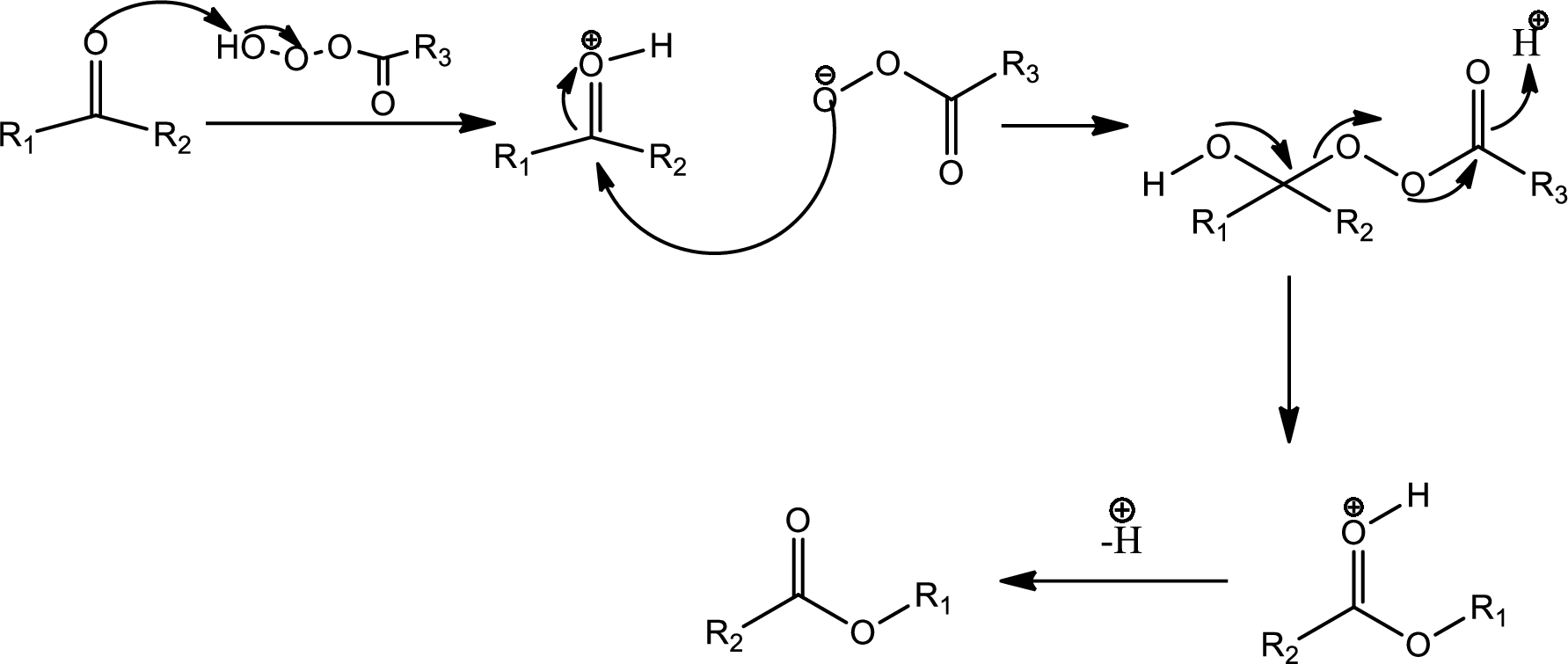
Baeyer-Villiger oxidation:
The Baeyer-Villiger oxidation is an organic rearrangement reaction that forms ester from a
(g)
Explanation of Solution
The reaction given in the question is,
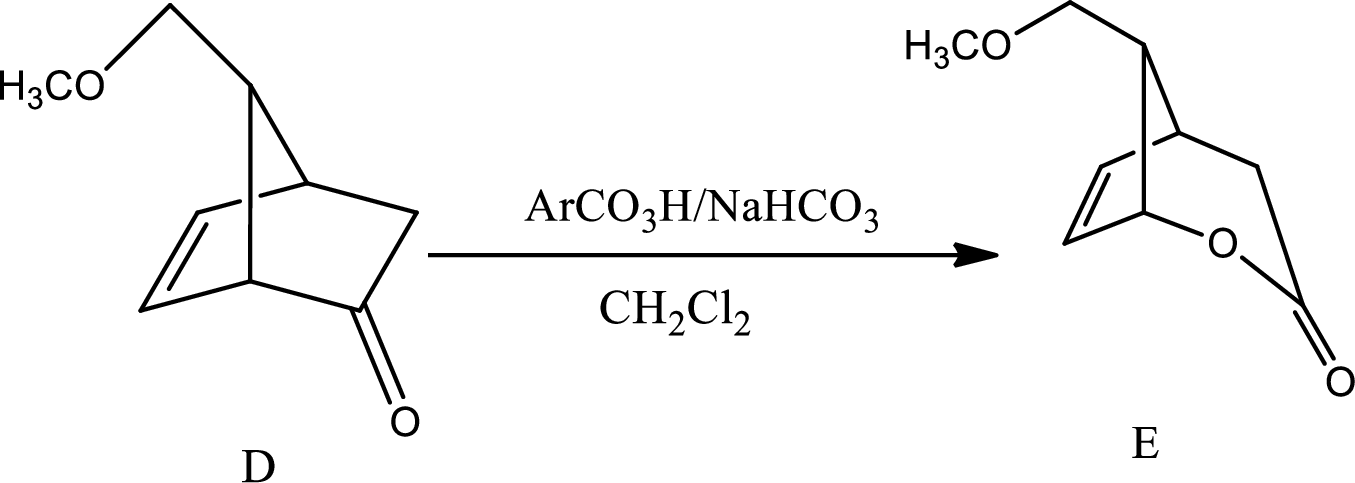
The mechanism is as follows,
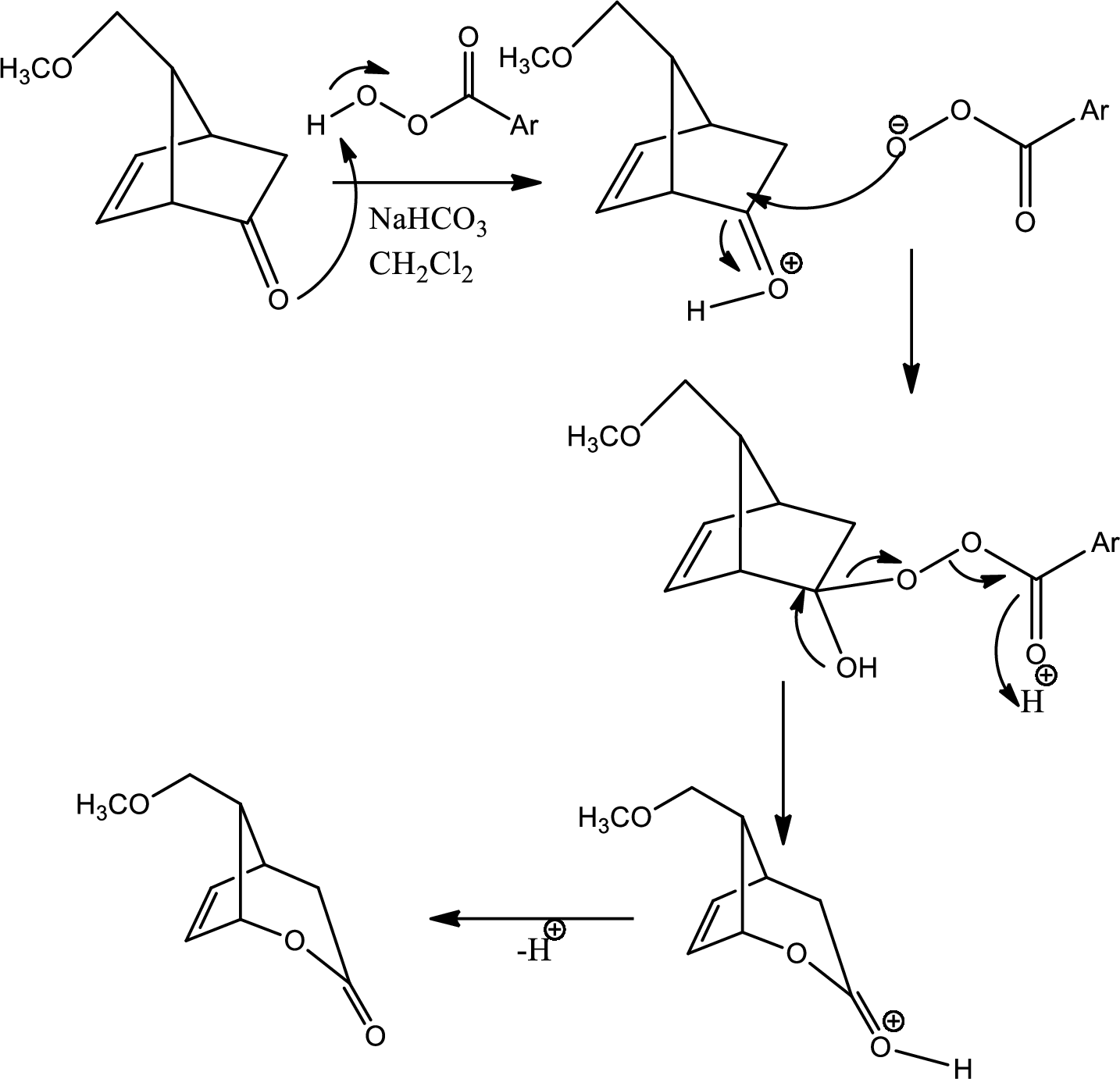
This is the pathway for D to E via Baeyer-Villiger oxidation. The process starts with acid base reaction followed by
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- Following is an outline of the stereospecific synthesis of the “Corey lactone.” Professor E. J. Corey (Harvard University) describes it this way. “The first general synthetic route to all the known prostaglandins was developed by way of bicycloheptene intermediates. The design was guided by the requirements that the route be versatile enough to allow the synthesis of many analogs and also allow early resolution. This synthesis has been used on a large scale and in laboratories throughout the world; it has been applied to the production of countless prostaglandin analogs. Note: The wavy lines in compound C indicate that the stereochemistry of -Cl and -CN groups was not determined. Q. You have not studied the Baeyer-Villiger reaction (D to E). The mechanism involves nucleophilic reaction of the peroxyacid with the carbonyl followed by a rearrangement much like that involved in the hydroboration reaction ). Write a mechanism for this reaction.arrow_forwardGamma(y)-amino butyric acid (GABA) is a neurotransmitter (a chemical that is used to send signals from one neuron to another) of the mammalian central nervous system. In order to understand how GABA works, conformationally restricted analogues, such as compound 1, have been made. During a synthesis of compound 1, compound 2 was subjected to allylic bromination using NBS and a radical initiator (AIBN) instead of light (Aust. J. Chem. 1981, 34, 2231-2236). H₂N. CH3 GABA CO₂H CH3 dddd CH3 H₂N Modify the structures given below to draw all eight possible allylic bromides that can be formed when compound 2 undergoes allylic bromination, considering all possible regiochemical and stereochemical outcomes. You can use the single bond tool to add/remove pi bonds. CH3 CO₂H CH3 solddd CH3 CH3 2 CH₂ CO₂Etarrow_forwardDiscovery of the antibiotic sulphanilamide led to rapid development of a large number of structurally related sulphonamides. Some of these were useful leads to compounds with other medicinal properties. Amongst these, sulfasalazine was active in the treatment of ulcerative colitis, a potentially fatal disease of the colon. As a medicinal chemist, you are about to carry out the synthesis of sulfasalazine, starting from aniline. Give the chemical names and draw the chemical structures of the reagents you will need to use for the all the steps marked (a)-(d). Aniline HO₂C HO (a) N=N- ACHN Ac represents CH3CO Sulfasalazine SO,NH S0,C1 N (b) (d) H₂N- SO,NH (c) N [Oro] SO,NH N Step (a) in question 5 above results in the para product only. Explain why this occurs.arrow_forward
- Following is an outline of the stereospecific synthesis of the “Corey lactone.” Professor E. J. Corey (Harvard University) describes it this way. “The first general synthetic route to all the known prostaglandins was developed by way of bicycloheptene intermediates. The design was guided by the requirements that the route be versatile enough to allow the synthesis of many analogs and also allow early resolution. This synthesis has been used on a large scale and in laboratories throughout the world; it has been applied to the production of countless prostaglandin analogs. Note: The wavy lines in compound C indicate that the stereochemistry of -Cl and -CN groups was not determined. Q. By what type of reaction is (B) converted to (C)?arrow_forward3 The 1H- and 13C-NMR data of an ester of molecular formula C6H10O2 are given below. Also shown are the COSY and HETCOR NMR spectra of the ester. Identify the ester, explaining how you reach your conclusion. 1H-NMR: 7.20-6.90 (1H), 5.85 (1H), 4.16 (2H), 1.88 (3H), 1.31 (3H) ppm 13C-NMR: 166.7, 144.5, 123.0 , 60.2, 18.0, 14.3 ppmarrow_forwardFollowing is a synthesis for toremifene, a nonsteroidal estrogen antagonist whose structure is closely related to that of tamoxifen. (a) This synthesis makes use of two blocking groups, the benzyl (Bn) group and the tetrahydropyranyl (THP) group. Draw a structural formula of each group and describe the experimental conditions under which it is attached and removed. (b) Discuss the chemical logic behind the use of each blocking group in this synthesis. (c) Propose a mechanism for the conversion of D to E. (d) Propose a mechanism for the conversion of F to toremifene. (e) Is toremifene chiral? If so, which of the possible stereoisomers are formed in this synthesis?arrow_forward
- Is it possible to distinguish between the following compounds using the spectroscopic techniques covered in CHE331? If so, discuss how. If not, discuss why.Benzonitrile vs Benzaldehyde(a) UV spectroscopy(b) IR spectroscopy(c) Mass spectrometry(d) 13C NMR spectroscopy(e) 1H NMR spectroscopyarrow_forwardAcid-catalyzed hydrolysis of HOCH2CH2C(CH3)2CN forms compound A (C6H10O2). A shows a strong peak in its IR spectrum at 1770 cm-1 and the following signals in its 1H NMR spectrum: 1.27 (singlet, 6 H), 2.12 (triplet, 2 H), and 4.26 (triplet, 2 H) ppm. Draw the structure for A and give a stepwise mechanism that accounts for its formation.arrow_forwardAddition of one equivalent of ammonia to 1-bromoheptane gives a mixture of heptan-1-amine,some dialkylamine, some trialkylamine, and even some tetraalkylammonium bromide.(a) Give a mechanism to show how this reaction takes place, as far as the dialkylamine.(b) How would you modify the procedure to get an acceptable yield of heptan-1-amine?arrow_forward
- Over the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Show how your proposals for compounds (2) and (3) can be converted to compound (1). (Note: In the course of developing this synthesis, Tietze discovered that vinylic bromides and iodides are more reactive in Heck reactions than are aryl bromides and iodides.)arrow_forwardOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q. In the course of the double Heck reactions, two new chiral centers are created. Assume in compound (3), the precursor to rings C and D of estrone, that the fusion of rings C and D is trans and that the angular methyl group is above the plane of the ring. Given this stereochemistry, predict the stereochemistry of…arrow_forwardOver the past several decades, chemists have developed a number of synthetic methodologies for the synthesis of steroid hormones. One of these, developed by Lutz Tietze at the Institut für Organische Chemie der Georg-August-Universität, Göttingen, Germany, used a double Heck reaction to create ring B of the steroid nucleus. As shown in the following retrosynthetic analysis, a key intermediate in his synthesis is compound (1). Two Heck reaction disconnects of this intermediate give compounds (2) and (3). Compound (2) contains the aromatic ring that becomes ring A of estrone. Compound (3) contains the fused five- and six-membered rings that become rings C and D of estrone. Q.Name the types of functional groups in estronearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

