
Concept explainers
Interpretation:
A model of naphthalene is given. The position of multiple bonds in it is to be shown. The number of possible resonance structures for naphthalene is to be stated and their structures are to be drawn.
Concept introduction:
Hydrogen is monovalent. Carbon is tetravalent and it can form four bonds. The position of double bonds can be identified by looking for carbons in the model having only three bonds. Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To determine:
The position of multiple bonds in the given model of naphthalene, C10H8. The number and structures of possible resonance structures for naphthalene are to be drawn
Answer to Problem 20VC
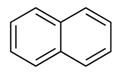
The positions of multiple bonds in naphthalene are shown in the structure given below.
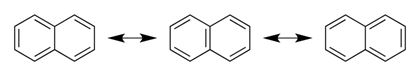
Naphthalene has three resonance forms as shown.
Explanation of Solution
In the model of naphthalene shown all the carbons have formed only three bonds and all the carbons require one more bond to satisfy their fourth valence. This fourth valence of each carbon can be satisfied by placing five double bonds between adjacent carbons as shown.
By shifting the position of double bonds, in total, three resonance structures can be drawn.
The positions of multiple bonds in naphthalene are shown in the structure given below.

Naphthalene has three resonance forms as shown.

Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
- Using the values of electronegativity given in Table 1.5, predict which indicated bond in each set is more polar, and using the symbols + and , show the direction of its polarity. (a) CH3OH or CH3OH (b) CH3NH2 or CH3 PH2 (c) CH3 SH or CH3SH (d) CH3F or HFarrow_forwardI need to indicate how the first resonance structure (the one with #4 above it) turns into the second one with arrows, but I cannot figure it out.arrow_forwardIn the following Lewis structure of [(CH3)2OH]+, every atom, bond and lone pair is positioned. To complete the structure, drag the formal charge tags to the appropriate atom(s). Each marker may be used more than once, or not at all. If an atom has a formal charge of zero, do not drag a tag to it. When you drag the marker in, place the little crosshairs in the upper left corner of the marker directly over the atom(s) in question (not above them). H H-C-O-C-H HHH - H I Η Η Η 0 0 + 2+ 2-arrow_forward
- HH NaH (base) D-CH3 2. Consider the following reaction: Draw 4 valid resonance structures/forms from this reaction:arrow_forwardIn the following Lewis structure of [(CH3)2OH]*, every atom, bond and lone pair is positioned. To complete the structure, drag the formal charge tags to the appropriate atom(s). Each marker may be used more than once, or not at all. If an atom has a formal charge of zero, do not drag a tag to it. When you drag the marker in, place the little crosshairs in the upper left corner of the marker directly over the atom(s) in question (not above them). H. H-C Н-С-О-С-Н C-H H HH 2- II 2-arrow_forwardIn the compound below, identify all carbon atoms that are electron deficient (8+) and all carbon atoms that are electron rich (d-). Justify your answer by drawing the correct resonance structures. H had H Harrow_forward
- 2. Draw the two resonance structures of benzene (nose 1q T)arrow_forward4. Draw two other reasonable resonance structures of this compound. Label each resonance form, including the one provided, as either "major" or "minor." :O: H3C H Harrow_forward2. Both pairs below show arrangements of the same (fictional) formula: A,X Which pair (a orb) represents resonance structures? Explain. a) A X=A A =X b) A X=A A XEA Aarrow_forward
- Label all lone pairs in the structures below. Also label how many degrees of unsaturation each molecule has. *HN= Are there any resonance forms for the structure above? If so, draw them below.arrow_forward1) Don't forget to show formal charges where necessary Draw the resonance hybrid for the following ion based upon the given resonance structures. C=CH₂ CH₂ Os. -CH₂arrow_forwardWhich of the following resonance structure for OCN- will contribute most to the correct structure of OCN-? O = C-N, with one lone pair on O and three lone pairs on N O=C=N, with two lone pairs on O, and two lone pairs on N O=C=N, with one lone pair on O, two lone pairs on C, and one lone pair on N O-C = N, with three lone pairs on O and one lone pair on N They All contribute equally to the structure on OCN-arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

