
Write a net ionic equation for any precipitation reaction that occurs when 1 M solutions of the following are mixed.
(a) copper(II) sulfate and sodium chloride
(b) manganese(II) nitrate and ammonium hydroxide
(c) silver nitrate and hydrochloric acid
(d) nickel(II) sulfate and potassium hydroxide
(e) ammonium carbonate and sodium nitrate
(a)
Interpretation:
The net ionic equation should be written when copper(II) sulfate and sodium chloride are mixed.
Concept introduction:
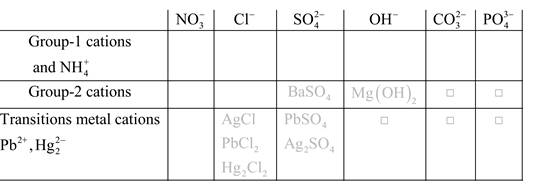
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
No precipitation occurs.
Explanation of Solution
Copper(II) sulfate:
Sodium chloride:
Reaction for the solution of copper(II) sulfate and sodium chloride is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
(b)
Interpretation:
The net ionic equation should be written when manganese(II) nitrate and ammonium hydroxide are mixed.
Concept introduction:
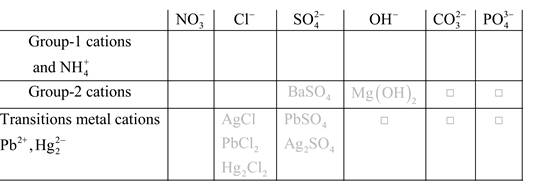
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
Precipitation occurs
The net ionic equation is:
Explanation of Solution
Manganese(II) nitrate:
Ammonium hydroxide:
Reaction for the solution of manganese(II) nitrate and ammonium hydroxide is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
So, the equation will be:
Now, cancelling out the ions which appear on both sides of the equation (
(c)
Interpretation:
The net ionic equation should be written when silver nitrate and hydrochloric acid are mixed.
Concept introduction:
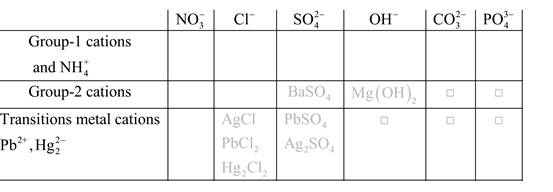
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
Precipitation occurs
The net ionic equation is:
Explanation of Solution
Silver nitrate:
Hydrochloric acid:
Reaction for the solution of silver nitrate and hydrochloric acid is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
So, the equation will be:
Now, cancelling out the ions which appear on both sides of the equation (
(d)
Interpretation:
The net ionic equation should be written when nickel(II) sulfate and potassium hydroxide are mixed.
Concept introduction:
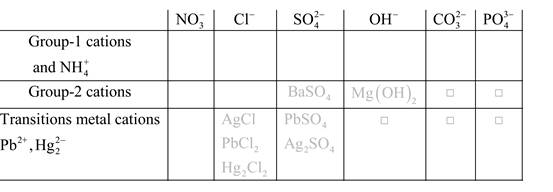
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
Precipitation occurs
The net ionic equation is:
Explanation of Solution
Nickel(II) sulfate:
Potassium hydroxide:
Reaction for the solution of silver nitrate and hydrochloric acid is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
So, the equation will be:
Now, cancelling out the ions which appear on both sides of the equation (
(e)
Interpretation:
The net ionic equation should be written when ammonium carbonate and sodium nitrate are mixed.
Concept introduction:
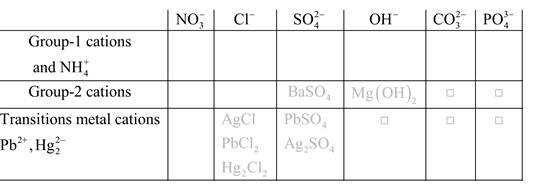
Solubility of any compound is predicted by above solubility chart.
Blank boxes indicate no precipitate formation occurs which means soluble in dilute solution.
Boxes with grey small box will form precipitate from dilute solutions and boxes where formula is written this is a cation-anion combination that will form precipitate.
Precipitation reactions: It is a type of chemical reactions where two soluble salts react with each other and formed different products, out of which one product must be insoluble in solution which is known as precipitate.
A chemical equation which shows only the species that are participated in the reaction is said to be net ionic equation.
Answer to Problem 9QAP
No precipitation occurs.
Explanation of Solution
Ammonium carbonate:
Sodium nitrate:
Reaction for the solution of silver nitrate and hydrochloric acid is written as:
Reactants:
Ions in solution:
Ions in solution:
Products:
Ions in solution:
Ions in solution:
Now,
Want to see more full solutions like this?
Chapter 4 Solutions
Chemistry: Principles and Reactions
- Arsenic acid, H3AsO4, is a poisonous acid that has been used in the treatment of wood to prevent insect damage. Arsenic acid has three acidic protons. Say you take a 25.00-mL sample of arsenic acid and prepare it for titration with NaOH by adding 25.00 mL of water. The complete neutralization of this solution requires the addition of 53.07 mL of 0.6441 M NaOH solution. Write the balanced chemical reaction for the titration, and calculate the molarity of the arsenic acid sample.arrow_forwardBone was dissolved in hydrochloric acid, giving 50.0 mL of solution containing calcium chloride, CaCL2. To precipitate the calcium ion from the resulting solution, an excess of potassium oxalate was added. The precipitate of calcium oxalate, CaC2O4, weighed 1.437 g. What was the molarity of CaCl2 in the solution?arrow_forwardIf aqueous solutions of potassium sulfide and iron(III) chloride are mixed, a precipitate is formed. Write the complete and net ionic equations for this reaction, and name the precipitate.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





