
Shown below is a diagram depicting the enthalpy change of a

- a Is the reaction exothermic or endothermic?
- b What is the sign of ΔH?
- c What is the sign of q?
- d If the reaction does no work, what is the sign of ΔE for this process?
(a)
Interpretation:
The nature of the given reaction has to be identified.
Concept introduction:
Exothermic reaction:
If heat is released during a chemical or physical change those reactions are called as exothermic reactions.
Endothermic reaction:
If heat is absorbed during a chemical or physical change those reactions are called as endothermic reactions.
Answer to Problem 6.32QP
Given reaction is endothermic reaction because the enthalpy increases moving from reactants to products.
Explanation of Solution
Given information,
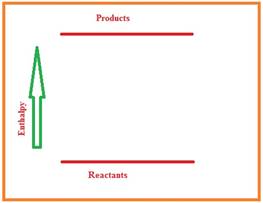
Figure 1
From the diagram we understand that the enthalpy of the reaction increases from reactants to products. Hence the given reaction is endothermic reaction.
(b)
Interpretation:
The sign of the enthalpy change
Concept introduction:
Exothermic reaction:
If heat is released during a chemical or physical change those reactions are called as exothermic reactions.
Endothermic reaction:
If heat is absorbed during a chemical or physical change those reactions are called as endothermic reactions.
Answer to Problem 6.32QP
The sign of enthalpy change
Explanation of Solution
Given information,

Figure 1
The enthalpy is increased from reactants to products so the sign of enthalpy is positive.
(c)
Interpretation:
The sign of heat
Concept introduction:
Exothermic reaction:
If heat is released during a chemical or physical change those reactions are called as exothermic reactions.
Endothermic reaction:
If heat is absorbed during a chemical or physical change those reactions are called as endothermic reactions.
Answer to Problem 6.32QP
The sign of
Explanation of Solution
Given information,

Figure 1
During the reaction the heat is absorbed so the sign of
(d)
Interpretation:
The sign of internal energy
Concept introduction:
Relationship between
The relationship between
Answer to Problem 6.32QP
The sign of internal energy change
Explanation of Solution
Given information,

Figure 1
Given information,
The reaction does no work.
The relationship between
Here, there is no pressure and volume work occurs, so the sign of internal energy change is positive
Want to see more full solutions like this?
Chapter 6 Solutions
General Chemistry - Standalone book (MindTap Course List)
Additional Science Textbook Solutions
Basic Chemistry
Fundamentals of Heat and Mass Transfer
Organic Chemistry
Chemistry: Matter and Change
Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
- The decomposition of ozone, O3, to oxygen, O2, is an exothermic reaction. What is the sign of q? If you were to touch a flask in which ozone is decomposing to oxygen, would you expect the flask to feel warm or cool?arrow_forwardWrite the balanced chemical equation for the combustion of methane, CH4(g), to give carbon dioxide and water vapor. Explain why it is difficult to predict whether S is positive or negative for this chemical reaction.arrow_forwardHow is the sign of q, heat, defined? How does it relate to the total energy of the system?arrow_forward
- The combustion of methane can be represented as follows: a. Use the information given above to determine the value of H for the combustion of methane to form CO2(g) and 2H2O(l). b. What is Hf for an element in its standard state? Why is this? Use the figure above to support your answer. c. How does H for the reaction CO2(g) + 2H2O (1) CH4(g) + O2(g) compare to that of the combustion of methane? Why is this?arrow_forwardCoal is used as a fuel in some electric-generating plants. Coal is a complex material, but for simplicity we may consider it to be a form of carbon. The energy that can be derived from a fuel is sometimes compared with the enthalpy of the combustion reaction: C(s)+O2(g)CO2(g) Calculate the standard enthalpy change for this reaction at 25C. Actually, only a fraction of the heat from this reaction is available to produce electric energy. In electric generating plants, this reaction is used to generate heat for a steam engine, which turns the generator. Basically the steam engine is a type of heat engine in which steam enters the engine at high temperature (Th), work is done, and the steam then exits at a lower temperature (Tl). The maximum fraction, f, of heat available to produce useful energy depends on the difference between these temperatures (expressed in kelvins), f = (Th Tl)/Th. What is the maximum heat energy available for useful work from the combustion of 1.00 mol of C(s) to CO2(g)? (Assume the value of H calculated at 25C for the heat obtained in the generator.) It is possible to consider more efficient ways to obtain useful energy from a fuel. For example, methane can be burned in a fuel cell to generate electricity directly. The maximum useful energy obtained in these cases is the maximum work, which equals the free-energy change. Calculate the standard free-energy change for the combustion of 1.00 mol of C(s) to CO2(g). Compare this value with the maximum obtained with the heat engine described here.arrow_forwardWhen a gas expands, what is the sign of w? Why? When a gas contracts, what is the sign of w? Why? What are the signs of q and w for the process of boiling water?arrow_forward
- The process of dissolving ammonium nitrate, NH4NO3, in water is an endothermic process. What is the sign of q? If you were to add some ammonium nitrate to water in a flask, would you expect the flask to feel warm or cool?arrow_forwardAthletic trainers use instant ice packs that can be cooled quickly on demand. Squeezing the pact breaks an inner container, allowing two components to mix and react. This reaction makes the pack become cold. Describe the heat flow for this spontaneous process.arrow_forwardThree gas-phase reactions were run in a constant-pressure piston apparatus as shown in the following illustration. For each reaction, give the balanced reaction and predict the sign of w (the work done) for the reaction. . If just the balanced reactions were given, how could you predict the sign of w for a reaction?arrow_forward
- 9.83 A student performing a calorimetry experiment combined 100.0 mL of 0.50 M HCl and 100.0 mL of 0.50 M NaOH in a coffee cup calorimeter. Both solutions were initially at 20.0°C, but when the two were mixed, the temperature rose to 23.2°C. (a) Suppose the experiment is repeated in the same calorimeter but this time using 200 mL of 0.50 M HCl and 200.0 mL of 0.50 M NaOH. Will the T observed he greater than, less than, or equal to that in the first experiment, and why? (b) Suppose that the experiment is repeated once again in the same calorimeter, this time using 100 mL of 1.00 M HCl and 100.0 mL of 1.00 M NaOH. Will the T observed he greater than, less than, or equal to that in the first experiment, and why?arrow_forwardWhen 1.000 g of gaseous butane, C4H10, is burned at 25C and 1.00 atm pressure, H2O(l) and CO2(g) are formed with the evolution of 49.50 kJ of heat. a Calculate the molar enthalpy of formation of butane. (Use enthalpy of formation data for H2O and CO2.) b Gf of butane is 17.2 kJ/mol. What is G for the combustion of 1 mol butane? c From a and b, calculate S for the combustion of 1 mol butane.arrow_forward9.53 Using these reactions, find the standard enthalpy change for the formation of 1 mol of PhO(s) from lead metal and oxygen gas. PbO(s)+C(graphite)Pb(s)+CO(g) H = 106.8 kJ 2C(graphite)+O2(g)2CO(g) H= -221.0 kJ If 250 g of lead reacts with oxygen to form lead(II) oxide, what quantity of thermal energy (in kJ) is ahsorhed or evolved?arrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





