
Concept explainers
(a)
Interpretation: The outcome of the indicated reaction should be predicted and explained mechanistically.
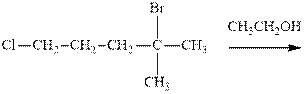
Concept introduction:Unimolecuar substitution or
A general

(b)
Interpretation: The outcome of the indicated reaction should be predicted and explained mechanistically.
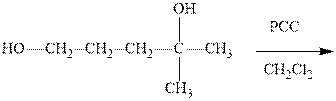
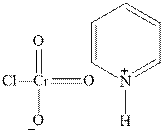
Concept introduction:PCC is pyridinium chlorochromate used as oxidizing reagent fop primary or secondary alcohols. The tertiary alcohols have no hydrogen so it cannot be oxidized. The structure of pyridinium chlorochromate is as follows:
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
Organic Chemistry: Structure and Function
- Provide a detailed mechanism for the following reaction:arrow_forward(4) Provide the reagents necessary to carry out the following synthesis. + enantiomer (A) (1) (i) BH3, (ii) H₂O2, NaOH; (2) NaOEt JATOT (B) (1) (i) BH3, (ii) H₂O2, NaOH; (2) TsCl, pyridine; (3) KOC(CH3)3 (C) (1) H3O+, (2) TsCl, pyridine, (3) KOC(CH3)3 (D) (1) HBr (no peroxides); (2) NaOEtarrow_forwardIllustrate the resonance effect of the methoxy group -OCH3, on the structure of the benzene ring. Draw all the oissuvke resonance forms of methoxybenzene, including the hybrid Based on the structures, explain how the presence of the -OCH3 group affects: (i) the reactivity of the benzene ring towards electrophilic attack (ii) the orientation or point of attack of an incoming electrophilic reagent on the benzene ring.arrow_forward
- Predict the major product of the following reactions. If it is possible, write all stereoisomers.arrow_forwardPropose and illustrate detailed mechanisms to complete the following reactionsarrow_forwardSuggest a detailed mechanism for each of the reactions below. Explain why the originating products were formed.arrow_forward
- gas, Cl¬CH2CH2¬S¬CH2CH2¬Cl, was used as a poisonous chemical agentin World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Itstoxicity stems from its ability to alkylate amino groups on important metabolic enzymes,rendering the enzymes inactive.(a) Propose a mechanism to explain why mustard gas is an exceptionally potent alkylatingagent.(b) Bleach (sodium hypochlorite, NaOCl, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes coloredcompounds to colorless compounds. Propose products that might be formed by thereaction of mustard gas with bleach.arrow_forwardPropose a reasonable reaction mechanism for the transformation given below and provide a brief explanationarrow_forwardPROPOSE A SUITABLE SYNTHESIS TO ACCOMPLISH THE FOLLOWING TRANSFORMATION.arrow_forward
- Synthesize the following compound from benzonitrile (C6H5CN): Please don't provide hand written solution...arrow_forwardPredict the major products of the following substitutions. Please provide explanations.arrow_forwardI'm having trouble proposing a syntheses for this molecule using the reatant given. Can you please help show how to approach this problem?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





