
Concept explainers
a)
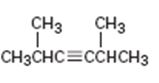
Interpretation:
The name of the
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the first of the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is given to it.
To give:
The name of the alkyne shown.
b)
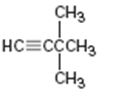
Interpretation:
The name of the alkyne shown is to be given.
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the first of the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is given to it.
To give:
The name of the alkyne shown.
c)
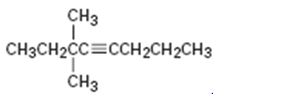
Interpretation:
The name of the alkyne shown is to be given.
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the first of the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is given to it.
To give:
The name of the alkyne shown.
d)
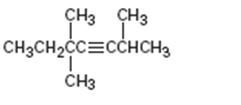
Interpretation:
The name of the alkyne shown is to be given.
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the first of the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is assigned to it.
To give:
The name of the alkyne shown.
e)
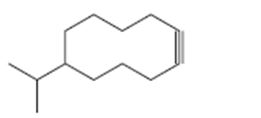
Interpretation:
The name of the alkyne shown is to be given.
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the first of the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is assigned to it.
To give:
The name of the alkyne shown.
f)
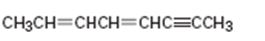
Interpretation:
The name of the alkyne shown is to be given.
Concept introduction:
The longest carbon chain which contains the carbon-carbon triple bond is chosen. The chain is numbered from the end that gives the lowest number to the carbon in triple bond. Compounds with more than one triple bond are called diynes, triynes and so forth. Compounds containing both double bond and triple bonds are called as enynes. The chain is numbered from the end nearer to the first of the multiple bonds, double or triple. When there is a choice in numbering the double bond is given preference and the lowest number is assigned to it.
To give:
The name of the alkyne shown.
Trending nowThis is a popular solution!

Chapter 9 Solutions
Organic Chemistry
- Name the following alkyne by an acceptable system of nomenclature:arrow_forwardDraw the structures of 5 structural nd positional isomers of alkynes with formula C6H10arrow_forwardn-o* transition can be found in these compounds: Alcohols and Aromatic Hydrocarbons. Alcohols and Amides Alkenes and Alkynes Alkynes and Aromatic Hydrocarbonsarrow_forward
- Which alkane most readily undergoes thermal decomposition? Note that C-H bonds are usually stronger than C-C bonds. O ethane O dimethylpropane O propane O methylpropanearrow_forwardhow to name cyclic or acyclic alkanes ? what are the steps and rules that I have to follow to name anyone that comes my wayarrow_forwardHow could 2,6-dimethylheptane be prepared from an alkyne and an alkyl halide? (The prime in R′ signifies that R and R′ can be different alkyl groups.)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





