
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 10, Problem 65P
When deuterated phenanthrene oxide undergoes a rearrangement in water to form a phenol, 81% of the deuterium is retained in the product.
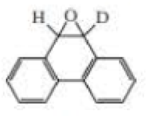
- a. What percentage of the deuterium is retained if an NIH shift occurs?
- b. What percentage or the deuterium is retained if an NIH shift does not occur?
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Identify the reagents you would use to achieve the following transformations:
Reagents 1
Reagents 2
Select from the given reagents:
A. Na, Pt
B. H2, NH3 (1) C. H2, Lindlar's cat.
D. Na, NH3 (I) E. H2, Pt
F. Na, Lindlar's cat.
Reagents 1
and Reagents 2
Which represents the correct arrow-pushing for the following transformation?
A.
B.
C.
D.
HeBr
H¬Br:
H¬Br:
Hor
Br
:Br:
What is the missing reagent in the sequence below?
A. HBr
B. Br₂ (dark)
C. Br2, FeBr3
D. Br₂, hn (or NBS)
O A
OB
O C
1. Zn(Hg), HCI
2. Missing reagent
3. NaOEt (E₂)
OD
Chapter 10 Solutions
Organic Chemistry (8th Edition)
Ch. 10.1 - Why are NH3 and CH3NH2 no longer nucleophiles when...Ch. 10.1 - Explain the difference in reactivity between...Ch. 10.1 - Prob. 5PCh. 10.1 - Prob. 6PCh. 10.1 - Prob. 8PCh. 10.2 - Prob. 9PCh. 10.3 - Prob. 11PCh. 10.3 - Show how 1-propanol can be converted into the...Ch. 10.4 - Which of the following alcohols dehydrates the...Ch. 10.4 - Prob. 14P
Ch. 10.4 - Prob. 15PCh. 10.4 - Propose a mechanism for each of the following...Ch. 10.4 - Draw the product of each of the following...Ch. 10.4 - Explain why the following alcohols, when heated...Ch. 10.4 - What stereoisomers are formed in the following...Ch. 10.4 - Prob. 20PCh. 10.4 - What alcohol would you treat with phosphorus...Ch. 10.5 - Prob. 22PCh. 10.6 - What are the major products obtained when each of...Ch. 10.6 - Prob. 26PCh. 10.7 - Prob. 27PCh. 10.7 - Would you expect the reactivity of a five-membered...Ch. 10.7 - Prob. 29PCh. 10.7 - What products are obtained from the reaction of...Ch. 10.7 - Prob. 31PCh. 10.7 - Prob. 32PCh. 10.7 - Prob. 33PCh. 10.8 - Draw the mechanism for formation of the two...Ch. 10.8 - Prob. 35PCh. 10.8 - Prob. 36PCh. 10.8 - How do the major products obtained from...Ch. 10.8 - Explain why the two arene oxides in Problem 38...Ch. 10.8 - Which compound is more likely to be carcinogenic?Ch. 10.8 - Three arene oxides can be obtained from...Ch. 10.9 - Explain why the half-life (the time it takes for...Ch. 10.10 - Prob. 43PCh. 10.10 - Prob. 44PCh. 10.10 - Prob. 45PCh. 10.10 - Prob. 46PCh. 10.10 - Prob. 47PCh. 10.10 - Describe a synthesis for each of the following...Ch. 10.11 - Using an alkyl halide and a thiol as starting...Ch. 10.11 - The following three nitrogen mustards were studied...Ch. 10.11 - Why is melphalan a good cancer drug?Ch. 10.11 - Prob. 53PCh. 10.12 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 55PCh. 10 - Which compound is more likely to be carcinogenic?Ch. 10 - Prob. 57PCh. 10 - Prob. 58PCh. 10 - When heated with H2SO4, both...Ch. 10 - What is the major product obtained from the...Ch. 10 - Write the appropriate reagent over each arrow.Ch. 10 - What alkenes would you expect to be obtained from...Ch. 10 - Prob. 63PCh. 10 - Prob. 64PCh. 10 - When deuterated phenanthrene oxide undergoes a...Ch. 10 - An unknown alcohol with a molecular formula of...Ch. 10 - Explain why the acid-catalyzed dehydration of an...Ch. 10 - Prob. 68PCh. 10 - Prob. 69PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - What product would be formed if the four-membered...Ch. 10 - Which of the following ethers would be obtained in...Ch. 10 - Using the given starting material any necessary...Ch. 10 - Prob. 74PCh. 10 - When 3-methyl-2-butanol is heated with...Ch. 10 - Draw structures for compounds AF.Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - How could you synthesize isopropyl propyl ether,...Ch. 10 - When ethyl ether is heated with excess HI for...Ch. 10 - When the following seven-membered ring alcohol is...Ch. 10 - Ethylene oxide reacts readily with HO because of...Ch. 10 - Describe how each of the following compounds could...Ch. 10 - Propose a mechanism for each of the following...Ch. 10 - Triethylene glycol is one of the products obtained...Ch. 10 - Prob. 85PCh. 10 - Propose a mechanism for the following reaction:Ch. 10 - Prob. 87PCh. 10 - An ion with a positively charged nitrogen atom in...Ch. 10 - The following reaction takes place several times...Ch. 10 - Prob. 90PCh. 10 - Propose a mechanism for each of the following...Ch. 10 - A vicinal diol has OH groups on adjacent carbons....Ch. 10 - Prob. 93PCh. 10 - Prob. 94PCh. 10 - Two stereoisomers are obtained from the reaction...Ch. 10 - Propose a mechanism for each or the following...Ch. 10 - Triethylenemelamine (TEM) is an antitumor agent....
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What is the best reagent(s) to carry out the following conversion? Br a. 1. Br2/FeBr3; 2. HNO3/H2SO4 b. 1. HNO3/H2SO4; 2. Br2/FeBr3 c. 1. NBS/peroxides; 2. HNO3/H2SO4 d. 1. H₂NCI/AICis; 2. H2/Pt; 3. Br2/FeBr3 Save Answer Question 14 of 32 What product would you expect in this multistep synthesis? 1. Br₂ hv 2. Li NO 2 3. Cul 4. bromocyclopentane/etherarrow_forward64. What reagent(s) will accomplish this conversion? a. EtMgBr followed by an acid wash b. EtLi followed by an acid wash Et2 CuLi followed by an acid wash с. d. Et3SiCl followed by an acid washarrow_forward11.-15. Match each reagent to how it reacts with Y is from the reagent. Answers may be repeated. 11. Br₂ 12. HSCH3 13. NaBH4_ 14. NH₂OH 15. (CH3)2Cu Lit HO. a. b. C. d. e. not a.-d.arrow_forward
- What major product will be formed when the shown Grignard reagent is combined with the shown organolithium reagent ? MgBr Li O1.C C O2B a 1:1 mixture of A and C 3. O 4. A O 5. Are you kidding us? Both the Grignard reagent and the organolithium are nucleophiles. They will not react with each other.arrow_forwardHelp me pleasearrow_forwardWhich set of reagents should be used to form the product shown below? O A. Heat, NaOH B. 1. HB(C6H11)2; 2. H202, NaOH O C. H20, H2S04, H9SO4 D. H20, H2SO4arrow_forward
- 2. Fill in the necessary reagents for each reaction. HO 1. A. В. 2.arrow_forwarda. HO b. NH 33. Which is a better nucleophile? a. H₂O or HO c. H₂S b. NH3 or NH₂ C. CH, CO in a better leaving group. or CH3CH₂O d. -0 or -0arrow_forwardBenzoyl chloride undergoes nucleophilic substitution at a faster rate than methyl propanoate because Select one: O a. the methoxide is a better leaving group than chloride. O b. chloride is a better leaving group than methoxide. O c. the acid chloride is more sterically hindered than the ester. O d. the ester is more sterically hindered than the acid chloride.arrow_forward
- The best reagents to carry out the reaction at right would be: a. CH₂ Hg(OAch H₂O NaBH4 KOII in CHCI. 2. Zn. II.O BH, THF H₂O₂, NaOH, H₂O CH₂ a™ OHarrow_forwardRank the compounds in each group according to their reactivity toward electrophilic substitution. 1 most reactive; 3 = least reactive. a. toluene methoxybenzene fluorobenzene b. benzonitrile ✓p-methylbenzonitrile ✓p-methoxybenzonitrilearrow_forwardWhich reagents are required to accomplish the transformation below? MeO A. 1. MeMgBr 2. H₂O B. 1. MeMgBr 2. MeBr C. NaBH4, MeOH D. 1. NaBH4, MeOH 2. NaH 3. MeBr MeO E. 1. LiAlH4 2. H₂O F. 1. LiAlH4 2. MeBr G. 1. H₂SO4 2. MeMgBr OMe + enant. H. 1. cat. TSOH, excess MeOH 2. LIAIH4 3. MeBrarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY