
Concept explainers
(a)
Interpretation:The complete Lewis structure of lisinopril needs to be drawn.
Concept Introduction: The complete Lewis structure of a molecule can be drawn using the total number of valence electrons present in it. It shows distribution of total number of valence electrons between the atoms of molecules. The bonded electrons are those electrons which are involved in bonding and they are represented as bonds or lines between the bonded atoms. The lone pair of electrons are those electrons which are not involved in bonding they are represented as dots (in pair) on the symbol of the atom.
(a)
Explanation of Solution
The given structure is as follows:
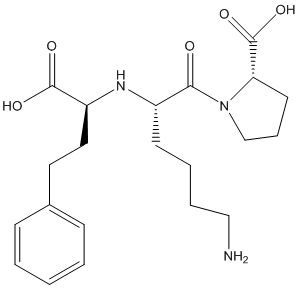
Here, O can have maximum of 2 lone pairs of electrons, N can have 1 lone pair of electrons and there is no lone pair of electrons on C and H atoms.
The valence electrons of C, H, N and O is 4, 1, 5 and 6 respectively.
The complete Lewis structure will be as follows:
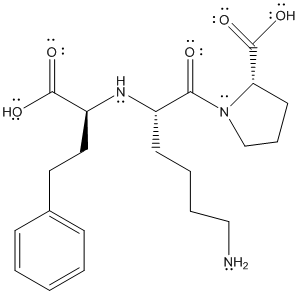
(b)
Interpretation:All the bond angles in lisinopril needs to be identified using the valence shell electron-pair repulsion model.
Concept Introduction: Hybridization is defined as mixing of orbital. The geometry of a central atom depends on its hybridization. The shape will be different from geometry if there are lone pair of electrons present on the central atom. The bond angles of central atom can be determined from its hybridization and geometry.
(b)
Explanation of Solution
The complete Lewis structure of lisinopril is as follows:
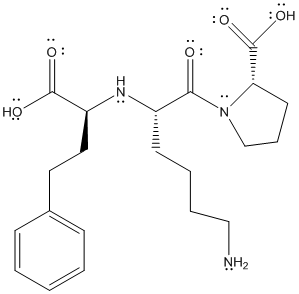
The valence electrons of C, H, N and O atom is 4, 1, 5 and 6 respectively. The C atom with 4 single bonds will have
The central atom with
(c)
Interpretation: The most polar bond in lisinopril needs to be determined.
Concept Introduction: A bond is said to be polar if there is electronegativity difference between the atoms. The most polar bond will be the one with maximum electronegativity difference between the bonded atoms.
(c)
Explanation of Solution
The structure is given as follows:
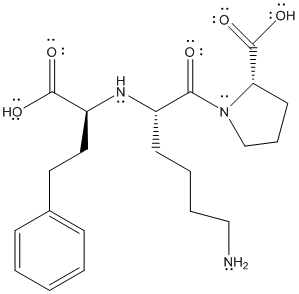
Here, C and H have comparable electronegativity thus, C-H bond is non-polar. The difference in electronegativity can be observed if C or H are bonded with O and N atoms.
Here, H is more electropositive than C thus, the electronegativity difference will be more in case of O-H or N-H bond as compared to O-C or N-C bonds.
Comparing O-H and N-H bond, O atom is more electronegative thus, electronegativity difference will be more in O-H and it is most polar bond in lisinopril.
(d)
Interpretation: Whether the given molecule is polar or non-polar needs to be determined.
Concept Introduction: If there are polar bonds present in a molecule then it is considered as a polar molecule.
(d)
Explanation of Solution
Due to the presence of polar O-H, N-H, C-O and C-N bonds, lisinopril is considered as a polar molecule.
(e)
Interpretation: This is to be explained whether lisinopril possess resonance or not.
Concept Introduction: Resonance is possessed by a molecule if there is possibility of delocalization of electrons. This can be positive if there is intercation between alternate lone pairs and pi bonds present in a molecule.
(e)
Explanation of Solution
The complete structure of lisinopril is as follows:
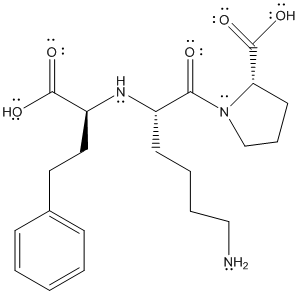
The above structure shows resonance as lone pair of electrons on oxygen atom is in conjugation with double bond. Similarly, lone pair of electrons on N atom is in conjugation with double bonded oxygen atom.
This is represented as follows:
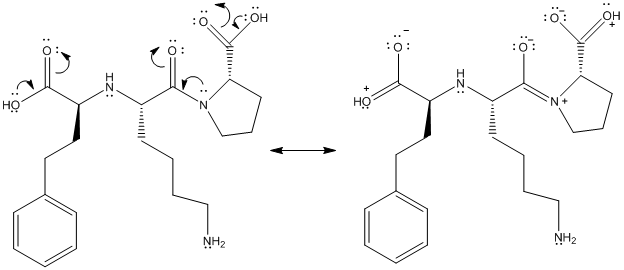
(f)
Interpretation: The
Concept Introduction: The functional groups are identity groups of the molecules. Functional groups define the types of
(f)
Explanation of Solution
The complete structure of lisinopril is as follows:
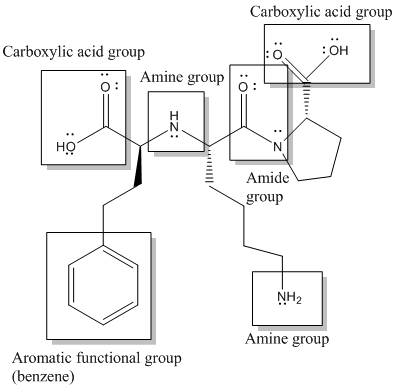
Here, -COOH, NH-, -NH2, CON- and benzene or
Functional groups are labelled as follows:
(g)
Interpretation: The molecular formula of lisinopril needs to be determined.
Concept Introduction: Molecular formula of any compound can be determined by calculating the total number of atoms of each element present in it.
(g)
Explanation of Solution
The complete structure of lisinopril is as follows:
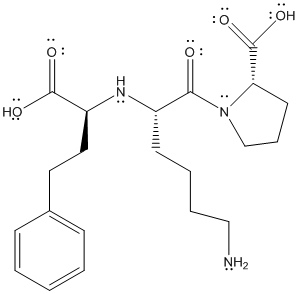
There are 4 types of atoms present in the above molecule namely carbon atom, oxygen atom, hydrogen atom and nitrogen atom.
From the above structure, the molecular formula will be
(h)
Interpretation: The types of intermolecular forces present in the lisinopril molecules needs to be determined.
Concept Introduction: Intermolecular forces are defined as type of forces present between the two molecules when they come in contact of each other. The type of forces depends on the physical and chemical properties of atoms present in the molecule.
(h)
Explanation of Solution
The complete structure is represented as follows:
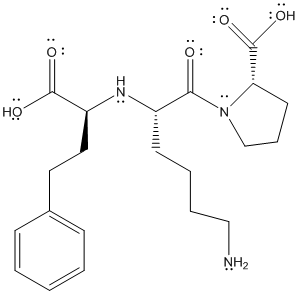
All the type of molecules containing non-polar bonds exhibits London dispersion forces. Due to the non-polar bonds (C-C and C-H) in the molecule. London dispersion forces are present.
Due to the presence of O-H bond, it also exhibits hydrogen bonding.
Want to see more full solutions like this?
Chapter 11 Solutions
Introduction To General, Organic, And Biochemistry
- The two compounds nitrogen dioxide and dinitrogen tetraoxide are introduced in Section 3.13. (a) NO2 is an odd-electron compound. Draw the best Lewis diagrams possible for it, recognizing that one atom cannot achieve an octet configuration. Use formal charges to decide whether that should be the (central) nitrogen atom or one of the oxygen atoms. (b) Draw resonance forms for N2O4 that obey the octet rule. The two N atoms are bonded in this molecule.arrow_forward3. Consider the four compounds BB33, CB14, NB13, and Br,O. Use periodic trends in bond lengths and the electronegativity table in Chang to answer the following questions: (a) Which molecule is expected to have the longest bonds? (b) Which molecule is expected to have the most polar bonds? (c) Which molecule is expected to have the least polar bonds?arrow_forwardshorter than the O-O bond in O2? Explain. 8.37 Which of the following statements about electronegativity is false? (a) Electronegativity is the ability of an atom in a molecule to attract electron density toward itself. (b) Electronegativity is the same thing as electron affinity. (c) The numerical values for electronegativity have no units. (d) Fluorine is the most electronegative element. (e) Cesium is the least electronegative element. 8,38 (a) What is the trend in electronegativity going fromarrow_forward
- 3) The molecule diphosphorus tetraoxide (P,O,) has two central atoms and four different resonance structures that do not violate the octet rule. Draw two of these resonance structures below. 4) The compound acetone is a common solvent. It has a chemical formula of CH,COCH, Acetone has three central atoms. (a) Draw the Lewis Dot structure for acetone. (b) Give the Ideal Bond Angle for all three central atoms. 5) Four covalent molecules are drawn below. :o: H. H-CH H H (1) (2) (3) (4) a) Define each of these molecules as polar or non-polar. (1) (2) (3) b) Describe the type of intermolecular force that each molecule would use: (1) (2) (3) (4)arrow_forwardWhich compounds have nonpolar covalent bonds, which have polar covalent bonds, and which have ions? (a) LiF (b) CH3F (c) MgCl2 (d) HClarrow_forwardConsider the formate ion, HCO2", which is the anion formed when formic acid loses an H* ion. The H and the two O atoms are bonded to the central C atom. (a) Draw the best Lewis structure(s) for this ion. (b) Are resonance structures needed to describe the structure? Explain briefly (c) Would you predict that the C-O bond lengths in the formate ion would be longer or shorter relative to those in CO2? Explain brieflyarrow_forward
- Consider the collection of nonmetallic elements O, P, Te,I, and B. (a) Which two would form the most polar singlebond? (b) Which two would form the longest single bond?(c) Which two would be likely to form a compound of formulaXY2? (d) Which combinations of elements would likelyyield a compound of empirical formula X2Y3?arrow_forward3) For each of the compounds MgO, Br2 and HBr:(a) What are the two atoms that formed each molecule?(b) What is their electronegativity difference between the atoms in each molecule?(c) What type of bond is formed in each molecule?(d) Are the electrons shared or transferred between the atoms in each molecule?arrow_forward(a) Use a polar arrow to indicate the polarity of each bond: N¬H, F¬N, I¬Cl. (b) Rank the following bonds in order of increasing polarity and decreasing percent ionic character: H¬N, H¬O, H¬C.arrow_forward
- Classify the following bonds as nonpolar, polar, or ionic: (a) the bond in ClF, (b) the bond in CsBr, and (c) the carbon-carbon double bond in C2H4.arrow_forward(1) What is the total number of valence electrons in the Lewis structure of SF2? electrons (2) Draw a Lewis structure for SF2arrow_forwardMethyl isocyanate, CH3NCO, was made infamous in 1984when an accidental leakage of this compound from a storagetank in Bhopal, India, resulted in the deaths of about3800 people and severe and lasting injury to many thousandsmore. (a) Draw a Lewis structure for methyl isocyanate.(b) Draw a ball-and-stick model of the structure,including estimates of all the bond angles in the compound.(c) Predict all the bond distances in the molecule.(d) Do you predict that the molecule will have a dipolemoment? Explain.arrow_forward
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
