
Concept explainers
(a)
Interpretation:
Electron dot structure for acrylonitrile and polyacrylonitrile and the rearranging of electron in the process of
Concept introduction:
The process that results in a final product of ceramic material from raw material is known as ceramic processing. The ceramic processing starts with fine powder of raw material combined by organic binder. Then it is shaped, compacted and at last sintered at temperature of 1300-2000 °C to decrease the voids between atoms and increase the density.
The strength and fracture toughness of ceramics is increased when fine grains of AL2O3 are reinforced by whiskers of SiC to form hybrid materials such as 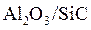 also known as ceramic composites.
also known as ceramic composites.
(b)
Interpretation:
The 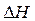 per
per 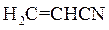 unit for conversion of acrylonitrile to polyacrylonitrile is to be calculated. Also, whether the reaction is exothermic or the endothermic should be determined.
unit for conversion of acrylonitrile to polyacrylonitrile is to be calculated. Also, whether the reaction is exothermic or the endothermic should be determined.
Concept introduction:
The enthalpy of reaction can be calculated by sum of
The expression to calculate 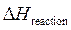 is as follows:
is as follows:
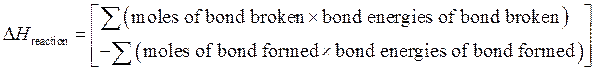
The negative sign in equation indicates that bonds will form in the products. It is an exothermic process, so the energy change is negative of bond energy.
Want to see the full answer?
Check out a sample textbook solution
Chapter 12 Solutions
CHEMISTRY-TEXT
- Teflon is prepared by the polymerization of tetra?uoroethylene. Write the equation that describes the polymerization using Lewis symbols.arrow_forwardExplain briefly why the tendency of a polymer to crystallize decreases with increasing molecular weightarrow_forward3. How does doping enhance the conducting properties of intrinsically conducting polymers? Explain with examples.arrow_forward
- Use an example to show how branching and crosslinking can affect the physical behavior of a polymer.arrow_forward(c) Compare and contrast the physical properties and mechanical behavior of typical polymers in the following classes of materials: (i) flexible plastics (ii) rigid plastics (iii) fibers (iv) elastomersarrow_forwardUse an example to show how branching and cross linking can affect the physical behavior of a polymer.arrow_forward
- Proteins are naturally occurring polymers formed by condensationreactions of amino acids, which have the generalstructureIn this structure, ¬R represents ¬H, ¬CH3, or anothergroup of atoms; there are 20 different natural amino acids,and each has one of 20 different R groups. (a) Draw the generalstructure of a protein formed by condensation polymerizationof the generic amino acid shown here. (b) When only afew amino acids react to make a chain, the product is called a“peptide” rather than a protein; only when there are 50 aminoacids or more in the chain would the molecule be called a protein.For three amino acids (distinguished by having three differentR groups, R1, R2, and R3), draw the peptide that resultsfrom their condensation reactions. (c) The order in which theR groups exist in a peptide or protein has a huge influence onits biological activity. To distinguish different peptides andproteins, chemists call the first amino acid the one at the “Nterminus” and the last one the one at…arrow_forwardPolymers that can conduct electricity are called conducting polymers. Some polymers can be made semiconducting; others can benearly metallic. Polyacetylene is an example of a polymer that is a semiconductor. It can also be doped to increase its conductivity.Polyacetylene is made from acetylene in a reaction that looks simple but is actually tricky to do:H—C≡ C— HAcetylene [— CH=CH— ]n Polyacetylene(a) What is the hybridization of the carbon atoms, and the geometry around those atoms, in acetylene and in polyacetylene?(b) Write a balanced equation to make polyacetylene from acetylene.arrow_forwardDuring our lectures on polymeric materials, I used polyethylene as an example because of its very straightforward structure. (a.) Hydrogen peroxide (H2O2) is used as an initiator for the polymerization of ethylene. Draw a diagram of a dissociated hydrogen peroxide molecule which emphasizes the creation of two free radicals (unbonded electrons) that can serve as initiators or as terminators of polymerization.arrow_forward
- What kind of chemical reaction do they have? is it Halogenation, Combustion, Oxidation or Polymerizationarrow_forwardIdentify any carbon atoms that change hybridization and the change in hybridization during the reactions in (a) 2-butene reacts with chlorine.(b) benzene burns in air.arrow_forwardWhy is it important to determine polymer chain length?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax





