
Concept explainers
In the mass spectrum of the following compounds, which is the tallest—the peak at m/z =57 or the peak at m/z = 71?
- a. 3·methylpentane
- b. 2·methylpentnne
Interpretation
The tallest peak should be identified.
Explanation of Solution
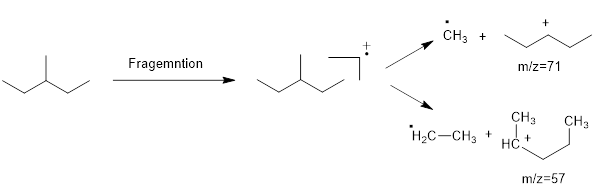
Mass fragmentation of 3-methyl pentane: In this molecule will be more adopted to lose an ethyl radical forming a secondary carbocation and a primary radical than a methyl radical forming secondary carbocation and methyl radical. In addition, 3-methylpentane ha undergoes for two path ways to lose an ethyl radical,
Therefore, the peak at
Mass fragmentation of 2-methyl pentane: The 2-methylpentane has undergoes for pathways to lose a methyl radical in each pathway and it cannot form a secondary carbocation by losing an ethyl radical. Loss of an ethyl radical would form a primary carbocation and a primary radical. Therefore, it will be more adopt to lose a methyl radical than an ethyl radical, the corresponding peak at
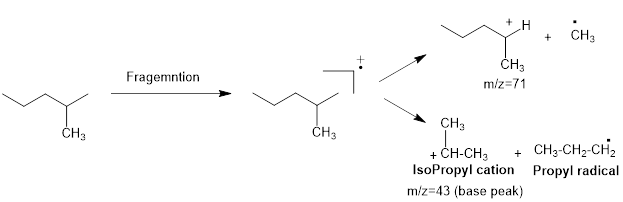
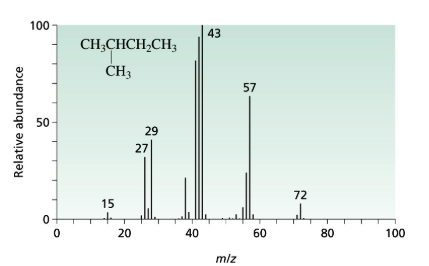
The 3-methyl pentane molecular peak at the m/e=71 has more intense then the peak at
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry; Organic Chemistry Study Guide A Format: Kit/package/shrinkwrap
- A) Label the mass spectrum of 1-butanol,3-methyl (Be sure to write the chemical formula for each major peak) (major peaks include 88, 70, 55, 42, and 43 m/z)arrow_forwardIs the compound giving rise to the mass spectrum shown here more likely to contain sulfur, bromine, or chlorine? Explain. 100 80 - 60- 40 20 40 80 120 160 m/z Relative abundance -유arrow_forwardIn the mass spectrum of an unknown organic compound, the molecular ion had a relative abundance of 19.0 while the M+1 had a relative abundance of 1.5. Estimate the number of carbons in the molecule. 1 4 7 2 LO 5 8 carbons 3 6 9 X Carrow_forward
- If the mass spectrum of a sample has a 7.7% M+1 peak compared to a 100% M+ peak, how many carbon atoms are we then likely to have in our chemical formula?arrow_forwardMass spectrometry is often used to both identify and quantify compounds. What is different between mass spectra with changing concentrations of a compound? How would you design a method (i.e. what would you need) to both identify and quantify a compound using mass spectrometry?arrow_forward2. In a mass spectrum of a compound, the following data (intensity) is given. M+ = 80, M+ +1 = 8.8, M+ +2 = 26. This compound will have carbons and will contain a a. 8, sulfur b. 9, silicon c. 10, chlorine d. 11, brominearrow_forward
- Which of the following formulae is consistent with a molecular ion of m/z 73 in a mass spectrometry experiment? O C3H8N2 O C4H11N O C4H10O O C3H5CIarrow_forwardWhat information is obtained from the mass spectrum and IRspectrum of an unknown compound X? Assume X contains theelements C, H, and O.arrow_forwardWhat molecular ions will be present in a mass spectrum of 2-chloropropane ? ( HINT: 1. Calculate the molecular weights using each of the common isotopes of Cl. 2. Write down the molecular formula for 2-choloropropane with the isotopes of chlorine and then calculate the mass of molecular ion , m/z).arrow_forward
- Draw the molecular ion (M*) for this molecule formed in the mass spectrometer. :0: Harrow_forwardWhat molecular ions will be present in a mass spectrum of 2-chloropropane,(CH3)2CHCl ?arrow_forwardA compound containing only carbon, nitrogen, oxygen, and hydrogen contains four carbon atoms. If the M+ peak in its mass spectrum appears at m/z = 87, then how many nitrogen atoms does it contain?arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

