
Concept explainers
Interpretation:
From, the given information, aufbau principle, needs to be explained.
Concept introduction:
Aufbau principle: According to Aufbau principle, the electron filling in different orbital takes place from lower to higher energy level. That is electron in lowest energy level fills before the highest energy level.
Answer to Problem 14STP
From the given electron configuration for silicon that is 1s2 2s2 2p63s2 3p2, it can be understand that is electron in lowest energy level fills before the highest energy level that is the Aufbau principle, which states that electron filling in different orbital takes place from lower to higher energy level.
Aufbau principle: According to Aufbau principle, the electron filling in different orbital takes place from lower to higher energy level. That is electron in lowest energy level fills before the highest energy level.
From the given electron configuration for silicon that is 1s2 2s2 2p63s2 3p2, it can be understand that is electron in lowest energy level fills before the highest energy level.
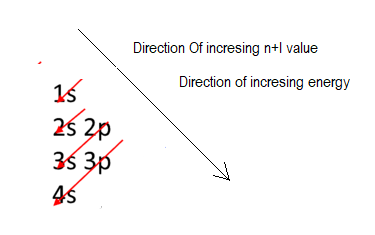
Energy of a level can be drived from its n+l value, which is lower for 1+0=1, than 2s which 2+0 and 2p which is 2+1.
That is electron first fill in 1s orbital then is 2s, which is higher in energy than 1s, after that electron fills in 2p which is higher in energy than 2s, then electron fills in 3s, which is higher in energy than 2p, after that electron fills in 3p which is higher in energy than 3s.
Explanation of Solution
Aufbau principle: According to Aufbau principle, the electron filling in different orbital takes place from lower to higher energy level. That is electron in lowest energy level fills before the highest energy level.
From the given electron configuration for silicon that is 1s2 2s2 2p63s2 3p2, it can be understand that is electron in lowest energy level fills before the highest energy level.
Energy of a level can be drived from its n+l value, which is lower for 1+0=1, than 2s which 2+0 and 2p which is 2+1.
That is electron first fill in 1s orbital then is 2s, which is higher in energy than 1s, after that electron fills in 2p which is higher in energy than 2s, then electron fills in 3s, which is higher in energy than 2p, after that electron fills in 3p which is higher in energy than 3s.
Chapter 14 Solutions
Chemistry: Matter and Change
Additional Science Textbook Solutions
Chemistry: A Molecular Approach (4th Edition)
Essential Organic Chemistry (3rd Edition)
Chemistry: Structure and Properties (2nd Edition)
Introductory Chemistry (6th Edition)
Chemistry: A Molecular Approach
General, Organic, and Biological Chemistry (3rd Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





