
Concept explainers
(a)
Interpretation:
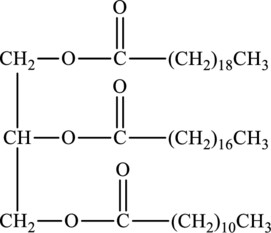
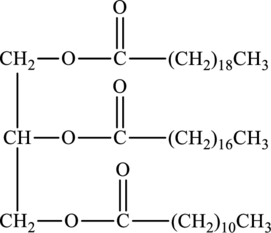
The fatty acids used to form below triacylglycerol have to be determined.
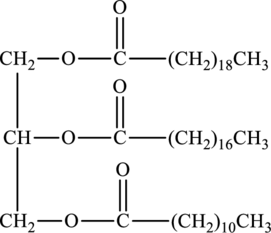
Concept Introduction:
Triacylglycerols are lipids that are formed by two or more similar or different fatty acids. They are also called triglycerides and are hydrophobic and nonpolar in nature. Addition of glycerol and three similar or different fatty acids results into triesters that is triacylglycerols. The general reaction for the synthesis of triacylglycerol is as follows:
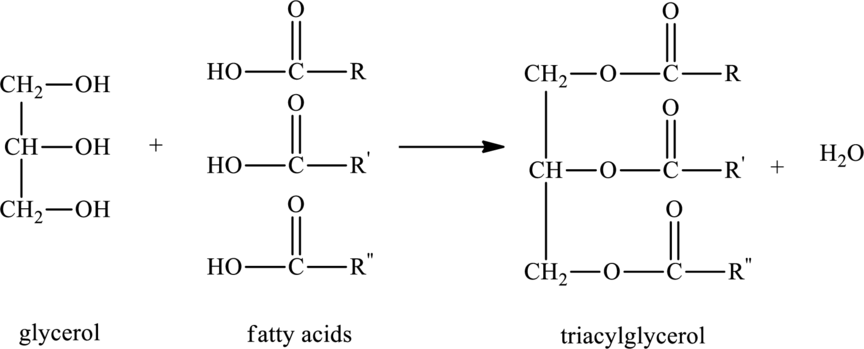
(b)
Interpretation:
Whether the below triacylglycerol is solid at room temperature or not has to be identified.
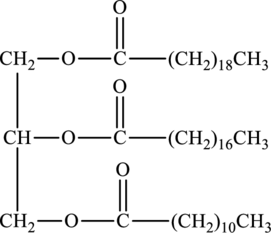
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
Hydrophobic region of below triacylglycerol has to be determined.
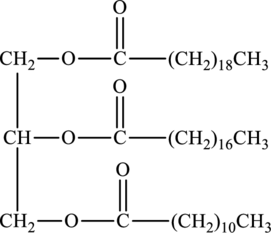
Concept Introduction:
Refer to part (a).
(d)
Interpretation:
Hydrophilic region of below triacylglycerol has to be determined.
Concept Introduction:
Refer to part (a).
(e)
Interpretation:
Product of hydrolysis of below triacylglycerol by aqueous sulfuric acid has to be determined.
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Principles of General, Organic, Biological Chemistry
- Give numerical answers to the following questions about the structure of a cholesterol molecule. a. How many six-membered rings arc present? b. How many amide linkages are present? c. How many hydroxyl substituents are present? d. How many total functional groups are present?arrow_forwardDraw structures of glyceryl tripalmitate and glyceryl trioleate. Which would you expect to have a higher melting point?arrow_forwardWhat functional group is common to all saponifiable lipids?arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div





