
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 58P
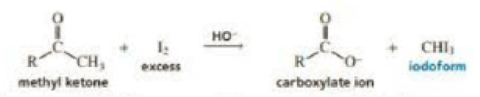
In the presence of excess base and excess halogen, a methyl ketone is converted to a carboxylate ion. The reaction is known as the haloform reaction because one of the products is haloform (chloroform, bromoform, or iodoform). Before spectroscopy became a routine analytical tool, the haloform reaction served as a test for methyl
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Please give me a step by step breakdown on how you get this answer.
How is nucleophilicity (nucleophile strength) related to basicity?
Amines are weak bases. Table 10.2 in the course textbook provides a list of pKb values for
several amines. The pK values for three amines and their structures are shown below. Explain
why increased substitution on nitrogen results in an increase of the pKb value.
H3C-NH2
PK = 3.36
H3C
N-H
pKb = 3.27
H₂C'
H3C
N-CH3
PK = 4.19
H₂C
Chapter 17 Solutions
Organic Chemistry (8th Edition)
Ch. 17.1 - Prob. 1PCh. 17.1 - Prob. 2PCh. 17.1 - Prob. 3PCh. 17.1 - Prob. 4PCh. 17.1 - Rank the compounds in each of the following groups...Ch. 17.2 - Explain why 92% of 2,4-pemtanedione exists as the...Ch. 17.3 - Draw the enol tautomers for each of the following...Ch. 17.3 - Prob. 8PCh. 17.4 - Prob. 9PCh. 17.4 - Prob. 10P
Ch. 17.5 - Show how the following compounds can be prepared...Ch. 17.6 - What compound is formed when a dilute solution of...Ch. 17.7 - Prob. 13PCh. 17.7 - Prob. 14PCh. 17.7 - How many stereoisomers are obtained from each of...Ch. 17.7 - Prob. 16PCh. 17.8 - Prob. 17PCh. 17.9 - Prob. 18PCh. 17.9 - What reagents should be used to prepare the...Ch. 17.10 - Prob. 20PCh. 17.10 - What aldehyde or ketone would be obtained when...Ch. 17.11 - Prob. 22PCh. 17.11 - How could you prepare the following compound using...Ch. 17.12 - Prob. 25PCh. 17.12 - What two carbonyl compound are required for the...Ch. 17.12 - Propose a mechanism for the following reaction:Ch. 17.13 - Draw the products of the following reactions:Ch. 17.13 - Prob. 29PCh. 17.13 - Prob. 30PCh. 17.14 - Prob. 31PCh. 17.15 - Write the mechanism for the reaction of a...Ch. 17.15 - Prob. 33PCh. 17.15 - Prob. 34PCh. 17.15 - Draw the product of the reaction of each of the...Ch. 17.16 - Draw the product obtained by heating each pair of...Ch. 17.16 - What two carbonyl compounds are needed to...Ch. 17.17 - Prob. 38PCh. 17.18 - Prob. 39PCh. 17.18 - Prob. 40PCh. 17.19 - Prob. 41PCh. 17.20 - Prob. 43PCh. 17.21 - Propose a mechanism for the formation of...Ch. 17.21 - Prob. 45PCh. 17.21 - a. If the biosynthesis of palmitic acid were...Ch. 17.21 - Prob. 47PCh. 17 - Prob. 48PCh. 17 - Prob. 49PCh. 17 - Number the following compounds in order of...Ch. 17 - The 1H NMR chemical shifts of nitromethane,...Ch. 17 - Prob. 52PCh. 17 - Draw the products of the following reactions: a....Ch. 17 - A racemic mixture of 2-methyl-1-phenyl-1-butanone...Ch. 17 - Draw the products of the following reaction:Ch. 17 - Prob. 56PCh. 17 - Prob. 57PCh. 17 - In the presence of excess base and excess halogen,...Ch. 17 - Identify A-L. (Hint: A shows three singles in its...Ch. 17 - Using cyclopentanone as the reactant, show the...Ch. 17 - Show how 4-methyl-3-hexanol can be synthesized...Ch. 17 - Show how the following compound can be synthesized...Ch. 17 - Show how the following compounds can be prepared...Ch. 17 - Prob. 64PCh. 17 - Prob. 65PCh. 17 - Indicate how each of the following compounds can...Ch. 17 - Prob. 67PCh. 17 - The ketone whose 1H NMR spectrum is shown here was...Ch. 17 - Indicate how the following compounds can be...Ch. 17 - Compound A with molecular formula C6H10 has two...Ch. 17 - Prob. 71PCh. 17 - Draw the products of the following reactions:Ch. 17 - Prob. 73PCh. 17 - a. Show how the amino acid alanine can be...Ch. 17 - Show how the following compounds can be...Ch. 17 - Prob. 76PCh. 17 - Explain why the following bromoketone forms...Ch. 17 - Prob. 78PCh. 17 - A carboxylic arid is formed when an -haloketone...Ch. 17 - An , -unsaturated carbonyl compound can be...Ch. 17 - What carbonyl compounds are required to prepare a...Ch. 17 - Prob. 82PCh. 17 - A Cannizzaro reaction is the reaction of an...Ch. 17 - Propose a mechanism for each of the following...Ch. 17 - The following reaction is known as the benzoni...Ch. 17 - Prob. 86PCh. 17 - Prob. 87PCh. 17 - Prob. 88PCh. 17 - Prob. 89PCh. 17 - Prob. 90PCh. 17 - Propose a mechanism for the following reaction:Ch. 17 - What reagents are required to convert the reactant...Ch. 17 - Starting with bromocyclohexane, how can each of...Ch. 17 - Describe how the following compounds can be...Ch. 17 - Prob. 4PCh. 17 - Describe three ways to synthesize the following...Ch. 17 - Explain why 92% of 2.4-pentanedione exists as the...Ch. 17 - Describe how the following compound can be...Ch. 17 - Prob. 8PCh. 17 - Prob. 9PCh. 17 - Prob. 10PCh. 17 - Show how the following compounds can be...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- predict the outcome of the reaction. explain briefly. answer letter c onlyarrow_forwardWrite chemical egs for the following reactions CH,NH, (CHJ,CH HO. HCI H,O ICH3 CHCH NH CHS NaOH H,O CHJ,CH NaOHarrow_forward1. Oxazolines (five membered ring with N and O shown below) are an important functional group in organic chemistry because they can be used to help facilitate catalytic, enantioselective reactions during the synthesis of important pharmaceutical molecules. One way to make oxazolines is through the ring expansion of N-acylaziridines. Me Ph Nal Acetone Ph An N-Acylaziridine An oxazoline a. This reaction utilizes nucleophilic substitution reactions to get from the aziridine to the oxazoline. Provide a complete arrow pushing mechanism for this transformation. You do not have to draw it while talking. You can have the mechanism already drawn out and simply talk through the steps. If you choose to use ChemDraw, be sure it is very clear where your arrows are pointing. You may also draw it by hand (paper or tablet) and screenshare an image of your work while explaining what happens. You must talk through each step of your mechanism and use proper terms such as nucleophile and electrophile. b.…arrow_forward
- Which of the following statements is true about the mechanism of acid-base reactions? The transition state has a tetrahedral A-H-B bond angle. The proton is shared only by B in the transition state. During the molecular collision in an acid-base reaction, the trajectory of the base, B-, must be toward the A of A-H. For an acid to transfer its proton to a base, the two entities must collide with specific geometry.arrow_forwardFor the Reaction: NH3+ H2O = NH4+ OH- When adding NH3+ phenolphthalien+ HCl What stress is applied and what direction does the equilibrium shift? When adding NH3+ phenolphthalien+ NH4Cl What stress is applied and what direction does the equilibrium shift? When adding NH3+ phenolphthalien+ NaOH What stress is applied and what direction does the equilibrium shift? When adding NH3+ phenolphthalien+ HCl What stress is applied and what direction does the equilibrium shift?arrow_forwardInspect the structure of the following compounds and explain which one will elute first andwhich one will be last to elute.arrow_forward
- At 25°C, for benzoate ion Kp = 1.54 x 10-10 while for ethanoate ion K = 5.5 x 10-10. Choose the correct answer: Select one: Ethanoate ion is a stronger base than benzoate ion but ethanoic acid is a weaker acid than benzoic acid. Ethanoate ion is a stronger base than benzoate ion but ethanoic acid is a stronger acid than benzoic acid. Ethanoate ion is a weaker base than benzoate ion and ethanoic acid is a weaker acid than benzoic acid. Ethanoate ion is a weaker base than benzoate ion but ethanoic acid is a stronger acid than benzoic acid.arrow_forwardPhenol (hydroxybenzene) behaves as a weak acid. a) Write out the equilibrium equation for its partial dissociation in water. b) Write out the expression for the acid dissociation constant, Ka. d) Draw the conjugate base of phenol and show how it is stabilised by resonance. e) Compare and explain the acidity of phenol (p = 9.9) with that of: cyclohexanol (pk = 16.0) 3-fluorophenol (pK₁ = 9.3) 4-acetylphenol (pK, = 8.1)arrow_forwardb) Rank the following molecules according to their acidity. Justify your answer. O OH NO₂ O OH CH 3 O OH CIarrow_forward
- Rank the following acids in decreasing acidity. Explain your answer CH2ClCOOH, CHCl2COOH, CH3COOHarrow_forwardWrite down the common (not IUPAC) names of the organic molecules that would be released if this molecule were hydrolyzed: CH₂ E CH CH,−O O || 11 O -(CH₂)7-CH=CH-CH₂-CH=CH-CH₂-CH=CH-CH₂-CH3 (CH₂)7 -CH=CH-CH₂-CH=CH-CH₂-CH=CH-CH₂-CH3 -(CH₂)16-CH3 Separate each name with a comma. You will find useful information in the ALEKS Data resource. a × Y Śarrow_forwardThe reaction of ozone with 2-butene leads to -10 formation of aldehyde + O ketones, aldehyde + alcohol, two molecules of carboxylic acid O.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY