
(a)
The PV diagram for each process in the cycle.
(a)
Answer to Problem 72P
The required PV diagram is shown in Figure 1.
Explanation of Solution
Given data:
The amount
The temperature
The pressure
The pressure
The temperature
The pressure
The temperature
The pressure
The temperature
The pressure
The temperature
Formula:
The expression to determine the initial volume of the gas is given by,
The volume of the gas after the adiabatic process is given by,
The expression for the temperature of the gas after adiabatic process is given by,
The volume of the gas after the isobaric process is given by,
The expression to determine the temperature of the gas after the isometric expansion is given by,
The expression for the volume of the gas after the isobaric process is given by,
Calculation:
The initial volume of the gas is calculated as,
The volume of the gas after the adiabatic process is calculated as,
The temperature of the gas after adiabatic process is calculated as,
Solve further as,
The volume of the gas after the isobaric process is calculated as,
The expression to determine the temperature of the gas after the isometric expansion is given by,
The expression for the volume of the gas after the isobaric process is given by,
From the above calculations the PV diagram for the different process is shown below.
The required diagram is shown in Figure 1
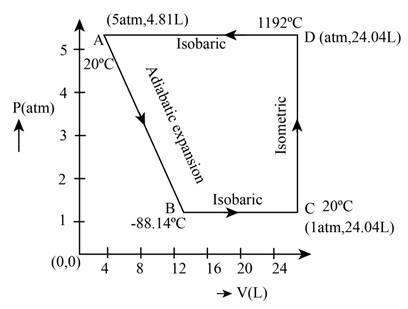
Figure 1
Conclusion:
Therefore, the required PV diagram is shown in Figure 1.
(b)
The work done by the gas during the complete cycle.
(b)
Answer to Problem 72P
The work done in the complete cycle is
Explanation of Solution
Formula Used:
The expression to determine the work done by the gas from
The expression to determine the work done by the gas from
The expression to determine the work done by the gas from
The expression for the total work during the complete cycle is given by,
Calculation:
The total work during the complete cycle is calculated as,
Solve further as,
Conclusion:
Therefore, the work done in the complete cycle is
(c)
The heat absorbed by the gas during the complete cycle.
(c)
Answer to Problem 72P
The heat absorbed by the gas is
Explanation of Solution
Formula:
The expression for the first law of
Calculation:
The heat absorbed by the gas is calculated as,
Conclusion:
Therefore, the heat absorbed by the gas is
Want to see more full solutions like this?
Chapter 18 Solutions
PHYSICS F/SCI.+ENGRS.,STAND.-W/ACCESS
- A sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P21.65). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state, (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally, (f) Find Q, W, and Eint for each of the processes, (g) For the whole cycle A B C A, find Q, W, and Eint.arrow_forwardA 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forward
- For a temperature increase of 10 at constant volume, what is the heat absorbed by (a) 3.0 mol of a dilute monatomic gas; (b) 0.50 mol of a dilute diatomic gas; and (c) 15 mol of a dilute polyatomic gas?arrow_forwardA gas in a cylindrical closed container is adiabatically and quasi-statically expanded from a state A (3 MPa, 2 L) to a state B with volume of 6 L along the path 1.8pV= constant. (a) Plot the path in the pV plane. (b) Find the amount of work done by the gas and the change in the internal energy of the gas during the process.arrow_forwardSuppose 26.0 g of neon gas are stored in a tank at a temperature of 152C. (a) What is the temperature of the gas on the Kelvin scale? (See Section 10.2.) (b) How many moles of gas are in the tank? (See Section 10.4.) (c) What is the internal energy of the gas? (See Section 10.5.)arrow_forward
- One of a dilute diatomic gas occupying a volume of 10.00 L expands against a constant pressure of 2.000 atm when it is slowly heated. If the temperature of the gas rises by 10.00 K and 400.0 J of heat are added in the process, what is its final volume?arrow_forwardFigure P21.45 shows a cyclic process ABCDA for 1.00 mol of an ideal gas. The gas is initially at Pi = 1.50 105 Pa, Vi = 1.00 103 m3 (point A in Fig. P21.45). a. What is the net work done on the gas during the cycle? b. What is the net amount of energy added by heat to this gas during the cycle? FIGURE P21.45arrow_forwardA 1.00-mol sample of hydrogen gas is heated at constant pressure from 300 K to 420 K. Calculate (a) the energy transferred to the gas by heat, (b) the increase in its internal energy, and (c) the work done on the gas.arrow_forward

 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning




