
(a)
The P - Vdiagram of the cycle, work done by the gas, heat absorbed by the gas and change in internal energy of the gas.
(a)
Answer to Problem 31P
The work done by the gas in each step are
Explanation of Solution
Given:
The initial pressure of the gas is
The initial volume of the gas is
The pressure at state 2 and 3 is
The volume at state 3 and 4 is
The pressure final pressure of the gas is
Formula used:
The expression for the specific heat ratio is given as,
Here,
The expression for heat absorbed by the gas in stage 1-2 is given as,
The expression for heat absorbed by the gas in stage 2-3 is given as,
The expression for heat absorbed by the gas in stage 3-4 is given as,
The expression for heat absorbed by the gas in stage 4-1 is given as,
The expression for the work done in stage 2-3 is given as,
The expression for the work done in stage 4-1 is given as,
The expression for change in internal energy in stage 1-2 is given as,
The expression for change in internal energy in stage 2-3 is given as,
The expression for change in internal energy in stage 3-4 is given as,
The expression for change in internal energy in stage 4-1 is given as,
Calculation:
The P-V diagram for the cycle can be given as,
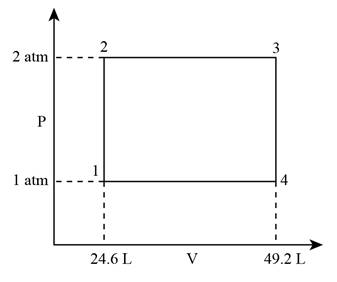
Figure 1
For monoatomic gas specific heat at constant volume and constant pressure can be given as,
The specific heat ratio can be calculated as,
The heat absorbed by the gas in stage 1-2 can be calculated as,
The expression for heat absorbed by the gas in stage 2-3 is given as,
The expression for heat absorbed by the gas in stage 3-4 is given as,
The expression for heat absorbed by the gas in stage 4-1 is given as,
The work done in stage 1-2 is zero because it is constant volume process.
The work done in stage 2-3 can be calculated as,
The work done in stage 3-4 is zero because it is constant volume process.
The work done in stage 4-1 can be calculated as,
The expression for change in internal energy in stage 1-2 is given as,
The expression for change in internal energy in stage 2-3 is given as,
The expression for change in internal energy in stage 3-4 is given as,
The expression for change in internal energy in stage 4-1 is given as,
Conclusion:
Therefore,the work done by the gas in each step are
(b)
The efficiency of the cycle.
(b)
Answer to Problem 31P
The efficiency of the cycle is
Explanation of Solution
Formula used:
The expression for total heat supplied to the cycle is given as,
The expression for total work done in the cycle is given as,
The expression for efficiency of the cycle is given as,
Here,
Calculation:
The total heat supplied to the cycle can be calculated as,
The total work done in the cycle can be calculated as,
The efficiency of the cycle can be calculated as,
Conclusion:
Therefore, the efficiency of the cycle is
Want to see more full solutions like this?
Chapter 19 Solutions
PHYSICS F/SCI.+ENGRS.,STAND.-W/ACCESS
- If a gas is compressed isothermally, which of the following statements is true? (a) Energy is transferred into the gas by heat. (b) No work is done on the gas. (c) The temperature of the gas increases, (d) The internal energy of the gas remains constant, (e) None of those statements is true.arrow_forwardA car tile contains 0.0380 m3 of air at a pressure of 2.20105 Pa (about 32 psi). How much more internal energy does this gas have than the same volume has at zero gauge pressure (which is equivalent to normal atmospheric pressure)?arrow_forwardWhen 400 J of heat are slowly added to 10 mol of an ideal monatomic gas, its temperature rises by 10 . What is the work done on the gas?arrow_forward
- A tank contains 111.0 g chlorine gas l2), which is at temperature 82.0 and absolute pressure 5.70105 Pa. The temperature of the air outside the tank is 20.0 . The molar mass of Cl2 is 70.9 g/mol. (a) What is the volume of the tank? (b) What is the internal energy of the gas? (c) What is the work done by the gas if the temperature and pressure inside the tank drop to 31.0 and 3.80105 Pa, respectively, due to a leak?arrow_forwardA mole of an ideal gas at pressure 4.00 atm and temperature 298 K expands isothermally to double its volume. What is the work done by the gas?arrow_forwardAn ideal gas initially at 300 K undergoes an isobaric expansion at 2.50 kPa. If the volume increases from 1.00 m3 to 3.00 m3 and 12.5 kJ is transferred to the gas by heat, what are (a) the change in its internal energy and (b) its final temperature?arrow_forward
- The insulated cylinder shown below is closed at both ends and contains an insulating piston that is flee to move on frictionless bearings. The piston divides the chamber into two compartments containing gases A and B. Originally, each compartment has a volume of 5.0102 m3 and contains a monatomic ideal gas at a temperature of and a pressure of 1.0 atm. (a) How many moles of gas are in each compartment? (b) Heat Q is slowly added to A so that it expands and B is compressed until the pressure of both gases is 3.0 atm. Use the fact that the compression of B is adiabatic to determine the final volume of both gases. (c) What are their final temperatures? (d) What is the value of Q?arrow_forwardTwo moles of a monatomic ideal gas at (5 MPa, 5 L) is expanded isothermally until the volume is doubled (step 1). Then it is cooled isochorically until the pressure is 1 MPa (step 2). The temperature drops in this process. The gas is now compressed isothermally until its volume is back to 5 L, but its pressure is now 2 MPa (step 3). Finally, the gas is heated isochorically to return to the initial state (step 4). (a) Draw the four pi-cresses in the pV plane. (b) Find the total work done by the gas.arrow_forwardOne of a dilute diatomic gas occupying a volume of 10.00 L expands against a constant pressure of 2.000 atm when it is slowly heated. If the temperature of the gas rises by 10.00 K and 400.0 J of heat are added in the process, what is its final volume?arrow_forward
- A gas in a cylindrical closed container is adiabatically and quasi-statically expanded from a state A (3 MPa, 2 L) to a state B with volume of 6 L along the path 1.8pV= constant. (a) Plot the path in the pV plane. (b) Find the amount of work done by the gas and the change in the internal energy of the gas during the process.arrow_forwardIt is found that, when a dilute gas expands quasi-statically from 0.50 to 4.0 L, it does 250 J of work. Assuming that the gas temperature remains constant at 300 K, how many moles of gas are present?arrow_forwardA gas at a pressure of 2.00 atm undergoes a quasi-static isobaric expansion from 3.00 to 5.00 L. How much work is done by the gas? `arrow_forward

 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning



