
Concept explainers
(a)
Interpretation: The product of the given proton transfers reaction is to be drawn. The acid and base in the starting material, and the conjugate acid and base in the products are to be stated.
Concept introduction: In proton transfer reaction, acid donate protons to from a conjugate base and a base accepts protons to from a conjugate acid. An acid base reaction is represented as,
Answer to Problem 2.40P
The products of the given proton transfer reaction are
Explanation of Solution
The given starting material
The products of the given proton transfer reaction are
(b)
Interpretation: The product of the given proton transfers reaction is to be drawn. The acid and base in the starting material, and the conjugate acid and base in the products are to be stated.
Concept introduction: In proton transfer reaction, acid donate protons to from a conjugate base and a base accepts protons to from a conjugate acid. An acid base reaction is represented as,
Answer to Problem 2.40P
The products of the given proton transfer reaction are
Explanation of Solution
The given starting material
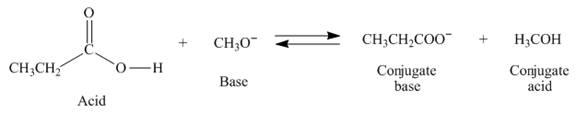
Figure 1
The products of the given proton transfer reaction are
(c)
Interpretation: The product of the given proton transfers reaction is to be drawn. The acid and base in the starting material, and the conjugate acid and base in the products are to be stated.
Concept introduction: In proton transfer reaction, acid donate protons to from a conjugate base and a base accepts protons to from a conjugate acid. An acid base reaction is represented as,
Answer to Problem 2.40P
The products of the given proton transfer reaction are
Explanation of Solution
The given starting material
The products of the given proton transfer reaction are
(d)
Interpretation: The product of the given proton transfers reaction is to be drawn. The acid and base in the starting material, and the conjugate acid and base in the products are to be stated.
Concept introduction: In proton transfer reaction, acid donate protons to from a conjugate base and a base accepts protons to from a conjugate acid. A acid base reaction is represented as,
Answer to Problem 2.40P
The products of the given proton transfer reaction are
Explanation of Solution
The given starting material
The products of the given proton transfer reaction are
Want to see more full solutions like this?
Chapter 2 Solutions
Organic Chemistry
- For the previous four questions, label each molecule that appears in the question or your answer asstrong acid, strong base, weak acid, or weak base.arrow_forwardWrite an equation to illustrate the equilibrium that is present when propanoic acid is dissolved in water. What structure predominates when OH is added to raise the pH to 12? What structure predominates as acid is added to lower the pH to 2?arrow_forward(E) Label each of the following as strong acid, strong base, or neither.arrow_forward
- a.What is the conjugate acid of A? b. What is the conjugate base of A?arrow_forward1) Circle the stronger acid of each pair. a. H₂Se or H₂S b. PH3 or H₂S c. H₂TeO₁ or H₂SO4 d. H₂SO4 or HCIO e. NH₂ or NH3arrow_forwardWhich one is the weakest base? A. aniline B. ammonia C. tetramethyl ammonium hydroxide D. ethylaminearrow_forward
- Answer the following questions about the four species A–D.a.Which two species represent a conjugate acid–base pair? b. Which two species represent resonance structures? c. Which two species represent constitutional isomers?arrow_forwardwhich of the following molecules would be the strongest base, and which would be the weakest base? NH A. B C D. a. C - strongest base, and D - weakest base b. C - strongest base, and B - weakest base C. A - strongest base, and D - weakest base d. D- strongest base, and A - weakest base e. B - strongest base, and Cweakest base f. D- strongest base, and C- weakest basearrow_forwardWhich of the following is the strongest acid? a. propane b. cyclopropane c. propene d. propyne a O b O darrow_forward
- 1. In the following acid-base reactions, the mechanism of reaction is shown. (i) Label the reactants as acid and base. (ii) Draw structures of the products in the appropriate boxes for conjugate acid and conjugate base. a. b. NH₂ 0:0: 1 Conjugate acid Conjugate acid + Conjugate base Conjugate basearrow_forwardb) Select the most acidic compound. H. H. B C A,arrow_forwardPyridine is a stronger base than pyrrole because the A. lone pair in pyridine is localized. B. lone pair in pyrrole is part of the continuous pi molecular orbital. C. lone pair in pyridine is more available for protonation. D. both A and B E. all of A, B, and Carrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



