
Concept explainers
a)
Interpretation:Molecular mass of the below-mentioned depiction is to be given. Also, correct name of compound should be done if it is named incorrectly.
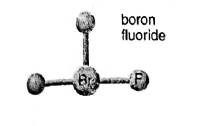
Concept introduction: In the molecular formula, the exact number and type of constituent atoms are shown. Type of constituent atoms is represented by their atomic symbols and number of each constituent atom is represented in the right subscript of its atomic symbol. In a hypothetical molecule
Formula to calculate molecular mass of
a)
Explanation of Solution
In the depiction three fluorine atoms are connected with central atom bromine. Hence the molecular formula of the molecules is
Molecular mass of
Name of the compound
b)
Interpretation: Molecular mass of the below-mentioned depiction is to be given. Also, correct name of compound should be done if it is named incorrectly.
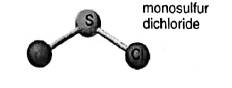
Concept introduction:In the molecular formula, the exact number and type of constituent atoms are shown. Type of constituent atoms is represented by their atomic symbols and number of each constituent atom is represented in the right subscript of its atomic symbol. In a hypothetical molecule
Formula to calculate molecular mass of
b)
Explanation of Solution
In the depiction two chlorine atoms are connected with central atom sulfur. Hence the molecular formula of the molecules is
Molecular mass of
Name of the compound
c)
Interpretation: Molecular mass of the below-mentioned depiction is to be given. Also, correct name of compound should be done if it is named incorrectly.
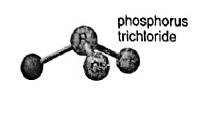
Concept introduction:In the molecular formula, the exact number and type of constituent atoms are shown. Type of constituent atoms is represented by their atomic symbols and number of each constituent atom is represented in the right subscript of its atomic symbol. In a hypothetical molecule
Formula to calculate molecular mass of
c)
Explanation of Solution
In the depiction three chlorine atoms are connected with central atom phosphorus. Hence the molecular formula of the molecules is
Molecular mass of
Name of the compound
d)
Interpretation: Molecular mass of the below-mentioned depiction is to be given. Also, correct name of compound should be done if it is named incorrectly.
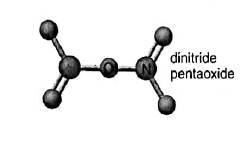
Concept introduction:In the molecular formula, the exact number and type of constituent atoms are shown. Type of constituent atoms is represented by their atomic symbols and number of each constituent atom is represented in the right subscript of its atomic symbol. In a hypothetical molecule
Formula to calculate molecular mass of
d)
Explanation of Solution
In the depiction two oxygen atoms are connected central nitrogen atom that forms
Molecular mass of
Name of the compound
Want to see more full solutions like this?
Chapter 2 Solutions
Principles of General Chemistry
- A compound associated with the odor of garlic on a persons breath has the chemical formula CyH8S and a formula mass of 88.19 amu. What number does y stand for in the chemical formula of the compound?arrow_forwardThe compound 1-propanethiol, which is the eye irritant released when fresh onions are chopped up, has the chemical formula C3HyS and a formula mass of 76.18 amu. What number does y stand for in the chemical formula of the compound?arrow_forwardSaccharin, a molecular model of which is shown below, is more than 300 times sweeter than sugar. It was first made in 1807, when it was common practice for chemists to record the taste of any new substances they synthesized. (a) Write the molecular formula for the compound, and draw its structural formula. (S atoms are yellow.) (b) If you ingest 125 mg of saccharin, what amount (moles) of saccharin have you ingested? (c) What mass of sulfur is contained in 125 mg of saccharin?arrow_forward
- Determine the molecular mass of the following compounds:arrow_forwardIn Groups 4A-6A, there are several elements whose symbols begin with S. Name these elements, and for each one give its symbol, atomic number, group number, and period. Describe each as a metal, metalloid, or nonmetal.arrow_forwardA small crystal of CaCl2 that weighs 0.12 g contains 6.5 1020 formula units of CaCl2. What is the total number of ions (cations and anions) that make up this crystal?arrow_forward
- Succinic acid (below) is an important metabolite in biological energy production. Give the molecular formula, molecular mass, and the mass percent of each element in succinic acid.arrow_forwardA chemist decomposes samples of several compounds; the masses of their constituent elements are shown. Calculate the empirical formula for each compound. 2.128 g Be, 7.557 g S, 15.107 g Oarrow_forwardThe molar masses and empirical formulas of several compounds containing carbon and chlorine are as follows. Find the molecular formula of each compound. 284.77 g/mol, CCl 131.39 g/mol, C2HCl3 181.44 g/mol, C2HClarrow_forward
- A chemist decomposes samples of several compounds; the masses of their constituent elements are listed. Calculate the empirical formula for each compound. 2.128 g Be, 7.557 g S, 15.107 g Oarrow_forwardab and Mastering 3 F1 2 F2 W Benadryl is an over-the-counter drug used to treat allergy symptoms. The formula of Benadryl is C17H₂1 NO. You may want to reference (Pages 217-220) Section 7.1 while completing this problem. #3 80 F3 E b My Questions | bartleby. $ 4 090 000 F4 R % 5 F5 openvellum.ecollege.com T ^ 6 How many moles of C are present in 0.732 moles of Benadryl? Express your answer to three significant figures and include the appropriate units Value Part B Submit Request Answer HÅ Submit Part C Value F6 μA How many moles of H are present in 2.35 moles of Benadryl? Express your answer to three significant figures and include the appropriate units Y MacBook Air E Course Home 1 P Pearson Copyright © 2022 Pearson Education Inc. All rights reserved. | Terms of Use | Privacy Policy | Permissions | Contact Us | & 7 Units Request Answer ଏ F7 U Units How many moles of N are present in 1.62 moles of Benadryl? Express your answer to three significant figures and include the appropriate…arrow_forwardGive the formula, name, and molecular mass of the following molecules:arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





