
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 41P
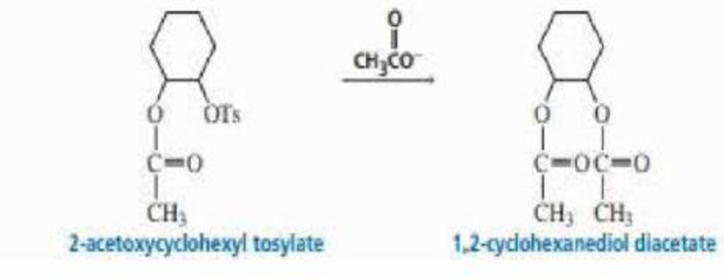
2-Acetoxycyclohexyl tosylate reacts with acetate ion to form 1,2-cyclohexanediol diacetate. The reaction is stereospecific—that is, the stereoisomers obtained as products depend on the stereoisomer used as a reactant. Recall that because 2-acetoxycyclohexyl tosylate has two asymmetric centers, it has four stereoisomers—two are cis and two are trans. Explain the following observations:
- a. Both cis reactants form an optically active trans product, but each cis reactant forms a different trans product.
- b. Both trans reactants form the same racemic mixture.
- c. A trans reactant is more reactive than a cis reactant.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
2-Acetoxycyclohexyl tosylate reacts with acetate ion to form 1,2-cyclohexanediol diacetate. The reaction is stereospecific—that is, the stereoisomers obtained as products depend on the stereoisomer used as a reactant. Recall that because 2-acetoxycyclohexyl tosylate has two asymmetric centers, it has four stereoisomers—two are cis and two are trans. Explain the following observations: a. Both cis reactants form an optically active trans product, but each cis reactant forms a different trans product. b. Both trans reactants form the same racemic mixture. c. A trans reactant is more reactive than a cis reactant
Under certain reaction conditions, 2,3-dibromobutane reacts with two equivalents of base to give three products, each of which contains two new p bonds. Product A has two sp hybridized carbon atoms, product B has one sp hybridized carbon atom, and product C has none. What are the structures of A, B, and C?
Draw the products formed when (S)-butan-2-ol is treated with TsCl and pyridine, followed by NaOH. Label the stereogenic center in each compound as R or S. What is the stereochemical relationship between the starting alcohol and the nal product?
Chapter 22 Solutions
Organic Chemistry (8th Edition)
Ch. 22.2 - Compare each of the mechanisms listed here with...Ch. 22.2 - Prob. 3PCh. 22.2 - Prob. 4PCh. 22.3 - a. Draw the mechanism for the following reaction...Ch. 22.5 - Prob. 7PCh. 22.5 - Propose a mechanism for the Co2+ catalyzed...Ch. 22.6 - Prob. 9PCh. 22.7 - Prob. 10PCh. 22.7 - Prob. 12PCh. 22.7 - Prob. 13P
Ch. 22.9 - Which of the following amino acid side chains can...Ch. 22.9 - Which of the following C-terminal peptide bonds is...Ch. 22.9 - Carboxypeptidase A has esterase activity as well...Ch. 22.10 - Arginine and lysine side chains fit into trypsins...Ch. 22.10 - Explain why serine proteases do not catalyze...Ch. 22.11 - If H2 18O is used in the hydrolysis reaction...Ch. 22.11 - Draw the pH-activity profile for an enzyme that...Ch. 22.12 - The pHactivity profile for glucose-6-phosphate...Ch. 22.12 - Prob. 23PCh. 22.13 - Draw the mechanism for the hydroxide ion-catalyzed...Ch. 22.13 - What advantage does the enzyme gain by forming an...Ch. 22.13 - Prob. 26PCh. 22.13 - Prob. 27PCh. 22.13 - Aldolase shows no activity if it is incubated with...Ch. 22 - Which of the following parameters would be...Ch. 22 - Prob. 29PCh. 22 - Prob. 30PCh. 22 - Prob. 31PCh. 22 - Indicate the type of catalysis that is occurring...Ch. 22 - The deuterium kinetic isotope effect (KH2O/KD2O)...Ch. 22 - Prob. 34PCh. 22 - Co2+ catalyzes the hydrolysis of the lactam shown...Ch. 22 - there are two kinds of aldolases. Class I...Ch. 22 - Prob. 37PCh. 22 - The hydrolysis of the ester shown here is...Ch. 22 - Prob. 39PCh. 22 - At pH = 12, the rate of hydrolysis of ester A is...Ch. 22 - 2-Acetoxycyclohexyl tosylate reacts with acetate...Ch. 22 - Proof that an imine was formed between aldolase...Ch. 22 - Prob. 43PCh. 22 - a. Explain why the alkyl halide shown here reacts...Ch. 22 - Triosephosphate isomerase (TIM) catalyzes the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Identify the stereochemical configuration of this cyclic compound. CI R Osarrow_forwardClassify the pair of compounds as the same compound, enantiomers, diastereomers, constitutional isomers, or not isomeric.. Also, select the correct IUPAC name, including the correct (R) or (S) designation, for each. CI CH3 H "Cl H Compound 1 The compounds are: enantiomers identical not isomeric diastereomers constitutional isomers H The correct IUPAC names are: H S... CI C/ CH3 Compound 2 Compound 1: (25,3R)-2,3-dichloropentane, Compound 2: (2S,3R)-2,3-dichloropentanearrow_forwardLabetalol has two chiral centers and, as produced in this synthesis, is a racemic mixture of the four possible stereoisomers. The active stereoisomer is dilevalol, which has the R,R configuration at its chiral centers. Draw a structural formula of dilevalol showing the configuration of each chiral center.arrow_forward
- Which of the following compounds are chiral? 1-Chloropropan-2-ol 2-Methylpropan-2-ol 2-Chloro-1-phenylpropane 2-Methylbutanal (A 1-Chloropropan-2-ol, 2-Methylpropan-2-ol, 2-Methylbutanal B 1-Chloropropan-2-ol,2-Methylpropan-2-ol C 1-Chloropropan-2-ol, 2-Methylpropan-2-ol, 2-Chloro-1-phenylpropane, 2-Methylbutanal D 1-Chloropropan-2-ol, 2-Chloro-1-phenylpropane, 2-Methylbutanalarrow_forwardName the following molecules, including all relevant stereochemical descriptors. 1 3 Br J 2 4 actionarrow_forwardHil H H C6H5-C-C-C6H5 C6H5 + HCI C6H5 H. ÓH ČI This equation shows the reaction of trans-2,3-diphenyloxirane with hydrogen chloride to form 2-chloro-1,2-diphenylethanol. How many total stereoisomers are possible for 2-chloro-1,2-diphenylethanol? 4 Given that opening of the epoxide ring in this reaction is stereoselective, provide the names of the only two isomeric products formed using IUPAC guidelines. product 1: (1R 2R) product 2: ((1S 2S) v 2-chloro-1,2-diphenylethanol 2-chloro-1,2-diphenylethanol Try Another Version 1 item attempt remaining Submit Answerarrow_forward
- Please provide IUPAC names for the given molecules, specifying their stereocentersarrow_forwardA Compound may have a possible number of which is H ₂ C ₁ stereoisomers formula 2^ (n = number of stereocentres. The compound (-) menthone (below) is a Natural Product found in peppermint. Use the given formula given to determine the number of possible stereoisomers in this Compound can have. I determined with the # CH(CH3)₂ H E 19arrow_forwardHow many stereocenters are there in the marine micro-algal toxin portimine? Label all stereocenters with an asterisk. Assign the stereochemistry in cases where this is unambiguously possible. N OH OH Iarrow_forward
- 4. What stereochemical descriptor is used to describe the stereochemistry in the following molecule? Br a) R b) S c) E d) Z e) cis f) transarrow_forwardIdentify the relationship between the given pairs of compounds. Choose from: A = enantiomers B = diastereomers C = constitutional isomers D = geometric isomers %3D E = conformational isomers F = two molecules of the same compound CO,H CH3 Br H H- Br H- Br H- Br CO,H CH3arrow_forward17) Specify the stereochemistry (R or S) at each of the following stereocenters. Write your answers in the boxes provides. Br H3C H •H • OH CH2CH3 SH ОНarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License