
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.3, Problem 13P
Using the pKa values in Section 2.3, rank the following species from strongest base to weakest base:
CH3NH2
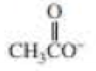
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
One of the three molecules is much more acidic than the other two. Identify the molecule
and explain why it is so much more acidic that the other two.
H
H
The C-H bond in acetone, (CH3)C=O, has a pką of 19.2. Draw two resonance structures for its
conjugate base. Then, explain why acetone is much more acidic than propane, CH;CH,CH3 (pKa = 50).
%3D
Which is the conjugate base?
C6H5NH2+H2O ↔ C6H5NH3+OH-
Chapter 2 Solutions
Organic Chemistry
Ch. 2.1 - Which of the following are not acids? CH3COOH CO2...Ch. 2.1 - Draw the products of the addbase renc1 ion when a....Ch. 2.1 - a. What is the conjugate acid of each or the...Ch. 2.2 - a. Which is a stronger acid: one with a pKa of 5.2...Ch. 2.2 - An acid has a Ka of 4.53 106 in water. What is...Ch. 2.2 - Prob. 6PCh. 2.2 - Antacids are compounds that neutralize stomach...Ch. 2.2 - Are the following body fluids acidic or basic? a....Ch. 2.3 - Draw the conjugate acid of each of the following:...Ch. 2.3 - a. Write an equation showing CH3OH reacting as an...
Ch. 2.3 - Estimate the pKa values of the following...Ch. 2.3 - a. Which is a stronger base: CH3COO or HCOO? (The...Ch. 2.3 - Using the pKa values in Section 2.3, rank the...Ch. 2.4 - Prob. 14PCh. 2.5 - a. For each of the acid-base reactions in Section...Ch. 2.5 - Ethyne has a pKa value of 25, water has a pKa...Ch. 2.5 - Which of the following bases can remove a proton...Ch. 2.5 - Calculate the equilibrium constant for the...Ch. 2.6 - Rank the ions (CH3, NH2, HO, and F) from most...Ch. 2.6 - Rank the carbanions shown in the margin from most...Ch. 2.6 - Which is the stronger acid?Ch. 2.6 - Prob. 22PCh. 2.6 - Rank the halide ions (F, Cl, Br, and l) from...Ch. 2.6 - a. Which is more electronegative, oxygen or...Ch. 2.6 - Which is a stronger acid? a. HCl or HBr b....Ch. 2.6 - a. Which of the halide ions (F, Cl, Br, and l) is...Ch. 2.6 - Prob. 27PCh. 2.7 - What is a stronger acid? a. CH3OCH2CH2OH or...Ch. 2.7 - Prob. 29PCh. 2.7 - What is a stronger base?Ch. 2.8 - Fosamax (shown on the previous page) has six...Ch. 2.8 - Which is a stronger acid? Why?Ch. 2.8 - Prob. 34PCh. 2.9 - Using the table of pKa values given in Appendix I,...Ch. 2.10 - Prob. 36PCh. 2.10 - As long as the pH is not less than _______, at...Ch. 2.10 - A naturally occurring amino acid such as alanine...Ch. 2.10 - For each of the following compounds, indicate the...Ch. 2.11 - Write the equation that shows how a buffer made by...Ch. 2.12 - Draw the products of the following react ions. Use...Ch. 2.12 - What product are formed when each of the following...Ch. 2 - a. Rank the following alcohols from strongest to...Ch. 2 - Which is a stronger base? a. HS or HO b. CH3O or...Ch. 2 - According to the explanations by Lewis, if a...Ch. 2 - a. Rank the following carboxylic acids from...Ch. 2 - Prob. 51PCh. 2 - For the following compound. a. draw its conjugate...Ch. 2 - Rank the following compounds from strongest to...Ch. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - a. Rank the following alcohols from strongest to...Ch. 2 - For each compound, indicate the atom that is most...Ch. 2 - a. Given the Ka values, estimate the pKa value of...Ch. 2 - A single bond between two carbons with different...Ch. 2 - Tenormin, a member of the group of drugs known as...Ch. 2 - From which of the following compounds can HO...Ch. 2 - a. For each of the following pairs of reactions,...Ch. 2 - Prob. 63PCh. 2 - Which is a stronger acid? a. b. c. d.Ch. 2 - Prob. 65PCh. 2 - Given that pH+ pOH = 14 and that the concentration...Ch. 2 - How could you separate a mixture of the following...Ch. 2 - Prob. 68PCh. 2 - a. If an add with a pKa of 5.3 is in an aqueous...Ch. 2 - Calculate the pH values of the following...Ch. 2 - Prob. 1PCh. 2 - Prob. 2PCh. 2 - Prob. 3PCh. 2 - Which of the reactions in Problem 3 favor...Ch. 2 - Prob. 5PCh. 2 - Prob. 6PCh. 2 - Prob. 7PCh. 2 - Which is the stronger acid? a. ClCH2CH2OH or...Ch. 2 - Prob. 9PCh. 2 - Prob. 10PCh. 2 - Which is a more stable base? Remembering that the...Ch. 2 - Which is the Stronger acid?Ch. 2 - Prob. 13PCh. 2 - a. Draw the structure of (CH3COOH (pKa = 4.7) at...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Rank the following substances in order of increasing acidity: (a) (CH3)2CHOH, HC≡CH, (CF3)2CHOH, CH3OH (b) Phenol, p-methylphenol, p-[trifluoromethyl) phenol (c) Benzyl alcohol, phenol, p-hydroxybenzonic acidarrow_forwardIn each equilibrium, label the stronger acid, the stronger base, the weaker acid, and the weaker base. Also estimate the position of each equilibrium. (a) CH3CH2O + CH3CCH CH3CH2OH + CH3CC (b) CH3CH2O + HCl CH3CH2OH + Cl (c) CH3COOH + CH3CH2O CH3COO + CH3CH2OHarrow_forwardRank the following in order of increasing basicity. The first in your list should be the least basic. HO 1 0 3 2 < 1 O2 3<1 01 2<3 01 3<2 3 1< 2 3. 2.arrow_forward
- The pKa of ascorbic acid (vitamin C, page 55) is 4.17, showing that it is slightly more acidic than acetic acid (CH3COOH, pKa 4.74). Compare the most stable conjugate base of ascorbic acid with the conjugate base of acetic acid, and suggest why these two compounds have similar acidities, even though ascorbic acid lacks the carboxylic acid (COOH) group.arrow_forwardRank the following species in order of increasing acidity. The least acidic should be on top, most acidic should be on the bottom. CFH2COOH CF2HCOOH CF3COOH CH3COOHarrow_forwardWhich is a stronger base? CH3 CHCO F CH3 CHCO- Brarrow_forward
- What is the conjugate acid of each of the following? What is the conjugate base of each? HNO2 H2Se HSe− H2C6H5O7− H3O2+arrow_forwardThe pKa of ascorbic acid (vitamin C, page 55) is 4.17, showing that it is slightly more acidic than acetic acid (CH3COOH, pKa 4.74).Compare the stabilities of these four conjugate bases, and predict which OH group of ascorbic acid is the most acidicarrow_forwardWhich of the acids below would have the strongest conjugate base? CH3CO2H pKa = 4.75 CICH2CO2H pKa = 2.81 C12CHCO2H pKa = 1.29 CH3CH2OH pKa = 18arrow_forward
- Consider the following acid-base equilibrium. HC6H₂O + C4H7O2¯= C6H50¯ + HC4H7O2 The position of the equilibrium lies to the left. Which compound is the weakest acid in this system? HC4H7O2 O C6H5O HC6H50 O C4H70₂arrow_forward2. Which of the following is most basic? Which is least basic? NH₂ WHECH 3arrow_forwardList the following alcohols in order from strongest acid to weakest acid. CH2=CHCH, OH, CH3 CH2 CH, OH, HC=CCH2OH Rank the following in order from strongest to weakest acid.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY