
Chemistry
3rd Edition
ISBN: 9780073402734
Author: Julia Burdge
Publisher: MCGRAW-HILL HIGHER EDUCATION
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.3, Problem 1PPC
Practice Problem CONCEPTUALIZE
CONCEPTUALIZE
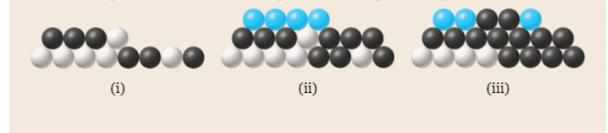
The following diagrams show collections of metal spheres. Each collection consists of two or more different types of metal—represented here by different colors. The masses of metal spheres are as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a Assume that mitochondria are cylinders 8.00 um in length and 0.800 pm in diameter.
What is the volume of a single mitochondrion?
The volume of a cylinder is given by the expression:
V = n x y2 x h
Volume=
m
Alina wants to make some yummy sugar cookies today !
The recipe written in her book is:
23/4 cup flour + 1 cup butter + 11/2 cup sugar+ 1 egg+1 tsp baking soda+ 1/2 tsp baking powder
= 24 sugar cookies.
But, Alina has the following ingredients in her kitchen: 9 cups flour, 5 cups butter, 4.5 cups sugar, 8 eggs and an excess of baking soda and powder.
She is very confused and is not sure as to how many cookies can she possibly make with this list. Can you help her?
1. How many cookies can she make?
2. Which ingredient is her Limiting reactant?
3. Besides, baking soda and powder , how much of each reactant will be left over in Alina's kitchen?
A typical "lead" pencil is actually made from carbon. If the carbon in the pencil weighs 1.50 gram
the pencil has (atoms] atoms of carbon.
How many copper atoms are in a pipe with a mass of 9.3 grams?
Chapter 2 Solutions
Chemistry
Ch. 2.1 - Practice Problem ATTEMPT
In each case, calculate...Ch. 2.1 - Practice Problem BUILD
(a) Two of the simplest...Ch. 2.1 - Practice Problem CONCEPTUALIZE
Which of the...Ch. 2.1 - For the two compounds pictured, evaluate the...Ch. 2.1 - Prob. 2CPCh. 2.2 - Practice ProblemATTEMPT How many protons,...Ch. 2.2 - Practice ProblemBUILD Give the correct symbols to...Ch. 2.2 - Prob. 1PPCCh. 2.3 - 2.3.1 How many neutrons are there in an atom of
Ch. 2.3 - Prob. 2CP
Ch. 2.3 - Practice ProblemATTEMPT The atomic masses of the...Ch. 2.3 - Practice ProblemBUILD The average atomic mass of...Ch. 2.3 - Practice Problem CONCEPTUALIZE
The following...Ch. 2.4 - Which of the following series of elemental symbols...Ch. 2.4 - 2.4.2 Which of the following elements would you...Ch. 2.4 - Practice ProblemATTEMPT Chloroform was used as an...Ch. 2.4 - Practice ProblemBUILD Write the molecular formula...Ch. 2.4 - Prob. 1PPCCh. 2.5 - Boron has two naturally occurring isotopes, which...Ch. 2.5 - 2.5.2 The two naturally occurring isotopes of...Ch. 2.5 - Practice Problem ATTEMPT
Name the following...Ch. 2.5 - Practice ProblemBUILD Name the following binary...Ch. 2.5 - Practice Problem CONCEPTUALIZE
Name the binary...Ch. 2.6 - Practice ProblemATTEMPT Give the molecular formula...Ch. 2.6 - Practice ProblemBUILD Give the molecular formula...Ch. 2.6 - Prob. 1PPCCh. 2.6 - Prob. 1CPCh. 2.6 - 2.7.2 What is the name of the compound...Ch. 2.6 - What is the correct formula for the compound...Ch. 2.6 - 2.7.4 What is the empirical formula of the...Ch. 2.7 - Prob. 1CPCh. 2.7 - Prob. 2CPCh. 2.7 - Prob. 3CPCh. 2.7 - 2.6.4 What is the formula of nickel(II) nitrate...Ch. 2.7 - Prob. 5CPCh. 2.7 - Prob. 6CPCh. 2.7 - Practice Problem ATTEMPT
Write empirical formulas...Ch. 2.7 - Practice ProblemBUILD For which of the following...Ch. 2.7 - Practice ProblemCONCEPTUALIZE Which of the...Ch. 2.8 - Practice Problem ATTEMPT
Name the following ionic...Ch. 2.8 - Practice Problem BUILD
Name the following ionic...Ch. 2.8 - Practice ProblemCONCEPTUALIZE The diagram...Ch. 2.9 - Practice Problem ATTEMPT
Deduce the formulas of...Ch. 2.9 - Practice ProblemBUILD Deduce the formulas of the...Ch. 2.9 - Practice Problem CONCEPTUALIZE
The diagram...Ch. 2.10 - Name the following species:...Ch. 2.10 - Name the following species:...Ch. 2.10 - Prob. 1PPCCh. 2.11 - Practice ProblemATTEMPT Determine the formula of...Ch. 2.11 - Practice ProblemBUILD Determine the formula of...Ch. 2.11 - Practice ProblemCONCEPTUALIZE Referring to the...Ch. 2 - Prob. 1KSPCh. 2 - Prob. 2KSPCh. 2 - Prob. 3KSPCh. 2 - What is the correct formula for phosphorus...Ch. 2 - What are the hypotheses on which Dalton's atomic...Ch. 2 - State the laws of definite proportions and...Ch. 2 - Prob. 3QPCh. 2 - Prob. 4QPCh. 2 - 2.5 Sulfur reacts with fluorine to produce three...Ch. 2 - 2.6 Both and contain only iron and oxygen. The...Ch. 2 - For the two compounds pictured, evaluate the...Ch. 2 - 2.8 For the two compounds pictured, evaluate the...Ch. 2 - Prob. 9QPCh. 2 - Prob. 10QPCh. 2 - Prob. 11QPCh. 2 - Describe the contributions of the following...Ch. 2 - 2.13 Describe the experimental basis for believing...Ch. 2 - The diameter of a neutral helium atom is about 1 ×...Ch. 2 - Prob. 15QPCh. 2 - Prob. 16QPCh. 2 - Prob. 17QPCh. 2 - Prob. 18QPCh. 2 - Prob. 19QPCh. 2 - 2.20 What is the mass number of an iron atom that...Ch. 2 - Prob. 21QPCh. 2 - 2.22 For each of the following species, determine...Ch. 2 - 2.23 Indicate the number of protons, neutrons, and...Ch. 2 - Prob. 24QPCh. 2 - Prob. 25QPCh. 2 - Prob. 26QPCh. 2 - Prob. 27QPCh. 2 - Prob. 28QPCh. 2 - What is the periodic table, and what is its...Ch. 2 - 2.30 State two differences between a metal and a...Ch. 2 - Prob. 31QPCh. 2 - Give two examples of each of the following: (a)...Ch. 2 - Prob. 33QPCh. 2 - Prob. 34QPCh. 2 - Prob. 35QPCh. 2 - Prob. 36QPCh. 2 - Prob. 37QPCh. 2 - Prob. 38QPCh. 2 - Prob. 39QPCh. 2 - Prob. 40QPCh. 2 - Prob. 41QPCh. 2 - Prob. 42QPCh. 2 - Prob. 43QPCh. 2 - The atomic masses of 1735Cl(75.53percent) and...Ch. 2 - The atomic masses of 204 Pb ( 1 .4 percent ) . 206...Ch. 2 - Prob. 46QPCh. 2 - Prob. 47QPCh. 2 - 2.48 What is the mass in grams of 13.2 amu?
Ch. 2 - Prob. 49QPCh. 2 - Prob. 71QPCh. 2 - Prob. 72QPCh. 2 - Prob. 73QPCh. 2 - Prob. 74QPCh. 2 - Prob. 75QPCh. 2 - Prob. 76QPCh. 2 - Prob. 77QPCh. 2 - Prob. 78QPCh. 2 - Prob. 79QPCh. 2 - Prob. 80QPCh. 2 - 2.61 Name the following compounds:
Ch. 2 - Prob. 82QPCh. 2 - Prob. 83QPCh. 2 - Prob. 84QPCh. 2 - Prob. 85QPCh. 2 - Prob. 86QPCh. 2 - Prob. 50QPCh. 2 - Prob. 51QPCh. 2 - Prob. 52QPCh. 2 - Prob. 53QPCh. 2 - Prob. 54QPCh. 2 - Prob. 55QPCh. 2 - Prob. 56QPCh. 2 - Prob. 57QPCh. 2 - Prob. 58QPCh. 2 - Prob. 59QPCh. 2 - Prob. 60QPCh. 2 - Prob. 61QPCh. 2 - Prob. 62QPCh. 2 - Prob. 63QPCh. 2 - Prob. 64QPCh. 2 - Prob. 65QPCh. 2 - Prob. 66QPCh. 2 - Prob. 67QPCh. 2 - Prob. 68QPCh. 2 - Prob. 69QPCh. 2 - Prob. 70QPCh. 2 - Prob. 87APCh. 2 - Prob. 88APCh. 2 - Prob. 89APCh. 2 - Prob. 90APCh. 2 - Prob. 91APCh. 2 - Prob. 92APCh. 2 - 2.93 What is wrong with or ambiguous about the...Ch. 2 - Prob. 94APCh. 2 - Prob. 95APCh. 2 - Prob. 96APCh. 2 - Prob. 97APCh. 2 - Prob. 98APCh. 2 - Prob. 99APCh. 2 - Prob. 100APCh. 2 - Prob. 101APCh. 2 - Prob. 102APCh. 2 - Prob. 103APCh. 2 - Determine the molecular and empirical formulas of...Ch. 2 - Prob. 105APCh. 2 - Prob. 106APCh. 2 - The Group 1B metals . Cu, Ag, and Au, are called...Ch. 2 - Prob. 108APCh. 2 - Prob. 109APCh. 2 - Prob. 110APCh. 2 - Two elements form a compound that can be...Ch. 2 - Which of the diagrams can be used to illustrate...Ch. 2 - Prob. 113APCh. 2 - Prob. 114APCh. 2 - Prob. 115APCh. 2 - 2.116 Show the locations of (a) alkali metals, (b)...Ch. 2 - Prob. 117APCh. 2 - Prob. 118APCh. 2 - Prob. 119APCh. 2 - 2.120 (a) Describe Rutherford’s experiment and how...Ch. 2 - Prob. 121APCh. 2 - Prob. 122APCh. 2 - Prob. 123APCh. 2 - A cube made of platinum (Pt) has an edge length of...Ch. 2 - Prob. 125APCh. 2 - Prob. 126APCh. 2 - Prob. 1SEPPCh. 2 - Prob. 2SEPPCh. 2 - Prob. 3SEPPCh. 2 - Prob. 4SEPP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which one of these are chemical change which ones are physical change? 1A) NaOH + Phenolphthalein 1B) NaOH + Phenolphthalein +HCl 2) NaH2CO3 +HC 3) CuSO4+ H2O 4) CuSO4 + NaOH 5) Mg + CuSO4 6) Mg + HCl 7) NH4Cl + H2Oarrow_forwardView Policies Current Attempt in Progress The structures of three compounds are shown below. Based on elemental composition of protein, which of the three compounds would you add to a food sample so that it would appear to contain more protein? Which of the three compounds would already be present in a food sample that actually did contain protein? +H3N-CH-Ĉ-0- NH2 CH2 N. H-C-OH CH2 CH,OPO}- H,N N. NH2 C=0 C O Adding compound B would significantly increase the apparent abundance of protein, while compound C would already be present in foods that actually contained protein. O Adding compound C would significantly increase the apparent abundance of protein, while compound B would already be present in foods that actually contained protein. O Adding compound B would significantly increase the apparent abundance of protein, while compound A would already be present in foods that actually contained protein. O Adding compound A would significantly increase the apparent abundance of protein,…arrow_forwardSTARTING AMOUNT X ADD FACTOR x( ).. 1.20 * 10²4 Determine the quantity of molecules in 2.00 moles of P4 2 g P4 INGRED 123.90 4 KUUZ Question 8 of 37 CERERE NARRA 1 2.00 molecules P4 ANSWER 6.022 x 1023 30.97 mol P4 RESET g/mol P4 5 2.41 x 10²4 4.82 x 10²4arrow_forward
- Also we have a mixture for gas is hydrogen helium neon and nitrogen sealed in a container of -50 Celsius degree so imaginary pick one molecule and follow it around for some time and record speed we determine that on average a small chill travels with speed of approximately 1.9x10^3 km/h . Which molecule or atom did we pick up MM,g/mol Hydrogen 2.02 Helium 4.00 Neon 20.18 Nitrogen 28.01arrow_forwardPriya brought a solid chocolate bar to school to eat as a snack in the afternoon. She left it in her pocket for a few hours. When she took out the chocolate bar to eat it, the bar was was no longer solid. Which description best describes the current state of the chocolate? A The chocolate is now made up of particles packed closely together, having both definite shape and definite volume. The chocolate is now made up of particles packed relatively close together, having an in- definite shape but a definite volume. C. The chocolate is now made up of particles that are far apart from each other, having both indefinite shape and indefinite volume. The chocolate is now made up of positively charged particles and negatively-charged electrons, having indefinite shape and indefinite volume.arrow_forwardA balance measures mass to 0.001 g. If you determine the mass of an object that weighs about 51 g, How would you record the mass? Question options: a) 51 g b) 51.1 g c) 51.08 g d) 51.075 g e) 51.0750 garrow_forward
- A piece of alminum foil contains 3.6 x 1021 aluminum atoms. 1) What is the mass of the aluminum foil in grams? 2) The density of aluminum is 2.7 g/cm3. What is the volume of the foil in cm3? 3) Rolls of aluminum foil are 310 mm wide and 0.016 mm thick (height). What length of foil in centimeters contains 3.6 x 1021 aluminum atoms?arrow_forwardArchimedes, a famous Greek scientist, was given a problem by King Hieron II of Syracuse (Sicily). The king suspected that his crown, which was supposed to be made of pure gold, contained some silver alloy, and he asked Archimedes to prove or disprove his suspicion. (It turned out that the crown did not contain silver.) How would you experimentally determined whether or not the crown was pure gold? (Hint: The method came to Archimedes when getting into a full bathtub. See the footnote in Experiment 22 for Archimedes’ solution.)arrow_forwardView History Bookmarks Develop Window Help A east.cengagenow.com [References) 1 pt EXERCISE Unit Conversion 1 pt Drag conversion units onto the boxes in the equation to make conversions. Some boxes can be left empty. Click on a unit to remove it from its position. 1 pt 1 cm³ Cu 9 g Cu 9.5 x 1021 atoms Cu 1 g Cu 1 pt 1 kg 1000 g 1 cm = 1 mL 1 pt 1 L 1000 cm3 1 pt 1 pt 520 L x 1 pt 1 pt 1 pt A piece of copper has a volume 520 L. What is the mass of the sample, in units of grams? In the boxes above, enter the correct setup that would be used to solve this problem. 1 pt 1 pt Check Next (2 of 3) 1 pt 1 pt Submit Answer Try Another Version 2 item attempts remaining 1 ptarrow_forward
- The mass of magnesium used in this experiment is critical to determining the moles of hydrogen gas generated. The analytical balances used in Chemistry 1A labs are very sensitive to point that touching materials with your fingers before measuring them can affect the measurements. The balances are sensitive enough to detect dirt and oils from your fingers transferred to the magnesium. Would your experimental value of R increase or decrease if the mass of magnesium measured was artificially high due the touching it with your hands? Increase Decreasearrow_forward2.) Suppose you're attempting to measure the density of a liquid that is highly volatile (it evaporates quickly). If you first measure the volume and then several minutes later you measure the mass of the liquid will the measured density be higher than the true value, lower than the true value, or the true density of the liquid. [It helps to understand this problem with the density equation.]arrow_forward10. Which of the following does not match the others? (A) 67.2 3.88 1000 100 (B) (C) 1s 2s 2p (D) ofelectronsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning
Measurement and Significant Figures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=Gn97hpEkTiM;License: Standard YouTube License, CC-BY
Trigonometry: Radians & Degrees (Section 3.2); Author: Math TV with Professor V;https://www.youtube.com/watch?v=U5a9e1J_V1Y;License: Standard YouTube License, CC-BY