
Concept explainers
(a)
Interpretation:
The structure of the functional group in alkene family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of
Alkenes are one of the important types of hydrocarbon which have at least one carbon-carbon double bond (
(b)
Interpretation:
The structure of the functional group in alcohol family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of functional groups are present and it includes alcohol, aldehyde, amine, ether, carboxylic acid etc.
Alcohol: It is an organic compound where it contains at least one
(c)
Interpretation:
The structure of the functional group in ester family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of functional groups are present and it includes alcohol, aldehyde, amine, ether, carboxylic acid etc.
Ester: Ester is a compound which is derived from an acid in which at least one hydroxyl group is replaced by a
(d)
Interpretation:
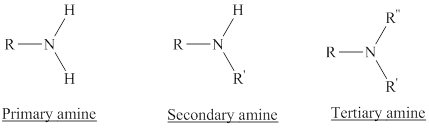
The structure of the functional group in amine family has to be given.
Concept introduction:
Functional group: They are certain substitutes in the organic molecules which determine the characteristic reactions taking place in it.
Different types of functional groups are present and it includes alcohol, aldehyde, amine, ether, carboxylic acid etc.
Amines: They are the derivatives of ammonia with one or more of the hydrogens replaced with a substituent such as an alkyl group.
Depending on the number of carbon side chain of the nitrogen, different types of amines can form.
Want to see the full answer?
Check out a sample textbook solution
Chapter 23 Solutions
General Chemistry: Atoms First
- Draw the condensed formulas for each of the following compounds:(a) dipropyl ether(b) 2,2-dimethyl-3-hexanol(c) 2-ethoxybutanearrow_forwardThe functional group of butanone is:(a) Carboxyl(b) ketonic(c) aldehydic(d) alcoholicarrow_forwardWrite down the distinguishing features among the following: (a) primary,secondary,tertiary Alcohols (b)primary, secondary, tertiary amines (c) carboxylic acid,ester and amide (d)Alkenes Alkynesarrow_forward
- Following is the structural formula of acetylsalicylic acid, better known by its common name aspirin. (a) Name the two oxygen-containing functional groups in aspirin. (b) What is the molecular formula of aspirin?arrow_forwardGive the IUPAC names of structures containing two carbon atoms for the following classes of compounds: (a) ether: (b) aldehyde: (c) carboxylic acid: (d) ester:arrow_forward(a) What is meant by the term isomer ? (b) Among the four alkanes, ethane, propane, butane, and pentane, which is capable of existing in isomeric forms?arrow_forward
- Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forward(a) Give the empirical formula and structural formula fora cyclic ether containing four carbon atoms in the ring.(b) Write the structural formula for a straight-chain compoundthat is a structural isomer of your answer to part (a).arrow_forward. (a) The insecticide methoprene (see Fig. 7.32d) is an ester. Write the structural formulas for the alcohol and the carboxylic acid that react to form it. Name the alcohol.(b) Suppose that the carboxylic acid from part (a) is changed chemically so that the OCH3 group is replaced by a hydrogen atom and the COOH group is replaced by a CH3 group. Name the hydrocarbon that would result.arrow_forward
- (b) Draw the structural formula for each of the following compounds. Lukiskan formula struktur bagi setiap sebatian berikut. (i) 2-iodo-4-methylpentan-3-amine (ii) 1,1-dibromo-2-chloro-2-methylbutane (iii) 2-ethyl-6-fluorophenolarrow_forwardGive an example reaction that would yield the following products. Name the organic reactant and product in each reaction. (a)alkane (b)monohalogenated alkanearrow_forward(a) The compound given below had the following IUPAC name and structural formula dibromocyclopentane C3H6CHBrCHBr (i) What type of isomerism is possible in the organic compound? (ii) Draw all the pairs of possible isomers and name them.arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College DivChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College DivChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


