
General Chemistry: Principles and Modern Applications (11th Edition)
11th Edition
ISBN: 9780132931281
Author: Ralph H. Petrucci, F. Geoffrey Herring, Jeffry D. Madura, Carey Bissonnette
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 24, Problem 65FP
Interpretation Introduction
Interpretation:
The oxidation states of Co ions in the following compound and the number of unpaired electrons (if the complex is low spin) should be determined also the structures of the two optical isomers should be drawn.
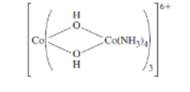
Concept introduction:
A special class of compounds is complex compounds in which the central atom(s)/ion(s) are bonded to the number of neutral or anion molecules. These compounds are also said to be a complex compound.
The bonded number of neutral or anion molecules to central metal is known as ligands that formally donate electrons to the metal center.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Werner prepared two compounds by heating a solution of PtCl₂ with triethyl phosphine, P(C₂H₅)₃, which is an excellent ligand for Pt. The two compounds gave the same analysis: Pt,38.8%; Cl, 14.1%; C, 28.7%; P, 12.4%; and H, 6.02%. Writeformulas, structures, and systematic names for the two isomers
An aqueous solution of copper sulfate (red line) was titrated with incremental amounts of a
ligand, forming a complex (blue lines). The UV-Vis spectra of this reaction are shown below:
2
330
380
430
480
530
580
630
680
730
780
Wavelength (nm)
What shift was observed and what can be said about the ligand's field strength?
O Hypsochromic shift caused by a weaker field ligand
O Bathochromic shift caused by a stronger field ligand
Hypsochromic shift caused by a stronger field ligand
Bathochromic shift caused by a weaker field ligand
Absorbance
When Alfred Werner was developing the field of coordinationchemistry, it was argued by some that the opticalactivity he observed in the chiral complexes he hadprepared was due to the presence of carbon atoms in themolecule. To disprove this argument, Werner synthesizeda chiral complex of cobalt that had no carbon atoms in it,and he was able to resolve it into its enantiomers. Design acobalt(III) complex that would be chiral if it could be synthesizedand that contains no carbon atoms. (It may not bepossible to synthesize the complex you design, but we willnot worry about that for now.)
Chapter 24 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Ch. 24 - Prob. 1ECh. 24 - Prob. 2ECh. 24 - Prob. 3ECh. 24 - Write appropriate formulas for the following. a....Ch. 24 - Prob. 5ECh. 24 - Prob. 6ECh. 24 - Prob. 7ECh. 24 - Prob. 8ECh. 24 - Prob. 9ECh. 24 - Prob. 10E
Ch. 24 - Prob. 11ECh. 24 - Prob. 12ECh. 24 - If A, B, C, and D are four different ligands, a....Ch. 24 - Prob. 14ECh. 24 - Prob. 15ECh. 24 - The structures of four complex ions are given....Ch. 24 - Prob. 17ECh. 24 - Prob. 18ECh. 24 - Prob. 19ECh. 24 - In contrast to the case of Co2+ considered in...Ch. 24 - Prob. 21ECh. 24 - Prob. 22ECh. 24 - Prob. 23ECh. 24 - Prob. 24ECh. 24 - Prob. 25ECh. 24 - Prob. 26ECh. 24 - Prob. 27ECh. 24 - Prob. 28ECh. 24 - Prob. 29ECh. 24 - Prob. 30ECh. 24 - Prob. 31ECh. 24 - Prob. 32ECh. 24 - Prob. 33ECh. 24 - Prob. 34ECh. 24 - Prob. 35ECh. 24 - Prob. 36ECh. 24 - Prob. 37ECh. 24 - Draw dashed and solid wedge diagrams of...Ch. 24 - Prob. 39IAECh. 24 - Prob. 40IAECh. 24 - Prob. 41IAECh. 24 - Prob. 42IAECh. 24 - Prob. 43IAECh. 24 - Prob. 44IAECh. 24 - Prob. 45IAECh. 24 - Prob. 46IAECh. 24 - Prob. 47IAECh. 24 - Prob. 48IAECh. 24 - Prob. 49IAECh. 24 - Prob. 50IAECh. 24 - Prob. 51IAECh. 24 - Prob. 52IAECh. 24 - Prob. 53IAECh. 24 - Prob. 54IAECh. 24 - The compound CoCl22H2O4NH2 may be one of the...Ch. 24 - Prob. 56IAECh. 24 - Provide a valence bond description of the bonding...Ch. 24 - Prob. 58IAECh. 24 - Prob. 59IAECh. 24 - Prob. 60IAECh. 24 - Prob. 61IAECh. 24 - Prob. 62IAECh. 24 - The graph that follows represents the molar...Ch. 24 - Prob. 64FPCh. 24 - Prob. 65FPCh. 24 - The crystal field stabilization energy (CFSE) can...Ch. 24 - In your own words, describe the following terms or...Ch. 24 - Briefly describe each of the following ideas,...Ch. 24 - Prob. 69SAECh. 24 - The oxidation state of Ni in the complex ion...Ch. 24 - Prob. 71SAECh. 24 - Prob. 72SAECh. 24 - Prob. 73SAECh. 24 - Prob. 74SAECh. 24 - Prob. 75SAECh. 24 - The most soluble of the following solids in...Ch. 24 - Prob. 77SAECh. 24 - Write appropriate formulas for the following...Ch. 24 - Prob. 79SAECh. 24 - Prob. 80SAECh. 24 - Prob. 81SAECh. 24 - Prob. 82SAECh. 24 - Prob. 83SAECh. 24 - Prob. 84SAE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A 3.03-g petroleum specimen was decomposed by wet ashing and subsequently diluted to 500 mL in a volumetric flask. Cobalt was determined by treating 25.00-mL aliquots of this diluted solution as follows: Assume that the Co(II)-ligand chelate obeys Beer’s law, and calculate the percentage of cobalt in the original sample.arrow_forwardCis-diamminedichloroplatinum (II) or “cisplatin” is a square planar complex with the formula [Pt(NH3)2Cl2]. Cisplatin is an anticancer drug that works by bonding to two adjacent guanine bases on the same strand of DNA, creating a kink which disrupts DNA replication. Each bond to a guanine base is formed by displacing one Cl ligand on cisplatin. The closely related complex trans-diamminedichloroplatinum (II) is completely ineffective as an anticancer agent. Explain why.arrow_forwardIdentify complexes that can have low and high spin configurations and rationalize why they adopt a particular configuration using a strong field / weak field ligand argument based on the spectrochemical series.arrow_forward
- Compare the methyl isocyanide (C=NCH3) ligand to C=O (carbonyl) with regards to its o donor ability and n-backbonding ability. Discuss. Clearly explain the origin of any differences in metal- ligand bonding between the two ligands.arrow_forwardIdentify complexes that can have low and high spin configurations and rationalize why they adopt a particular configuration using a strong field / weak field ligand argument based on the spectrochemical series. Specifically: know where CO,CN-,Cl-,Br¯,I¯,and NH3 fall on the spectrochemical series and why.arrow_forwardWhat is the most likely mechanism for ligand substitution in square-planar complexes? Group of answer choices association dissociation A wide variety of mechanisms are observed. Chemists have not determined one to take place in most cases. reductive eliminationarrow_forward
- What do you understand by ‘denticity of a ligand’?arrow_forwardThe oxalate ligand, [C:O.]², is a bidentate ligand and the structure of a complex ion [Fe(ox):]- is shown below. The view of diagram (b) below is along the O-Fe-O axis. Determine the point group of the complex ion and explain why molecules that belong to your determined point group are chiral. (a) (b)arrow_forwardDraw a Lewis structure for the cyclopentadienyl ligand (C5H5)- and explain why it is more likely to bind it’s aromatic protons to a transition metal center compared to the similar benzene molecule (C6H6).arrow_forward
- I. II. 2) Show how the d orbital splitting changes as an octahedral complex undergoes tetragonal distortion and eventually becomes a square planar complex. Describe the nephelauxetic effect. How does this effect explain the contribution of covalent bonding in metal-ligand bondarrow_forwardDetermine the hybridization of the metal centre in the complexes below using Ligand Field Theory. Give explanations to the hybridization that happened.arrow_forwardA transition metal, M, is coordinated in an octahedral fashion to the following ligands: Two chloro ligands; One (2-aminoethyl)phosphine ligand with the following structure abbreviation: N P One (0-CHz-CHz–SJ²-ligand with the following structure abbreviation: S a) Sketch all possible isomers, clearly indicating pairs of enantiomers b) Classify the configuration of chiral isomers as A or Aarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning



Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning