
Interpretation:
Using benzene and any other necessary
Concept introduction:
The steps involved in the synthesis of sulfathiazole from benzene are i) Nitration of benzene ii) Reduction of nitrobenzene iii) Acetylation of the amino group iv) Chlorosulfonation of the anilide v) Treatment with 2-aminothiazole vi) Hydrolysis of the anilide to an amine.
To Show:
How to synthesize sulfathiazole starting from benzene and using any other necessary amines.
Answer:
The synthesis of sulfathiazole starting from benzene and using any other necessary amines is given below.
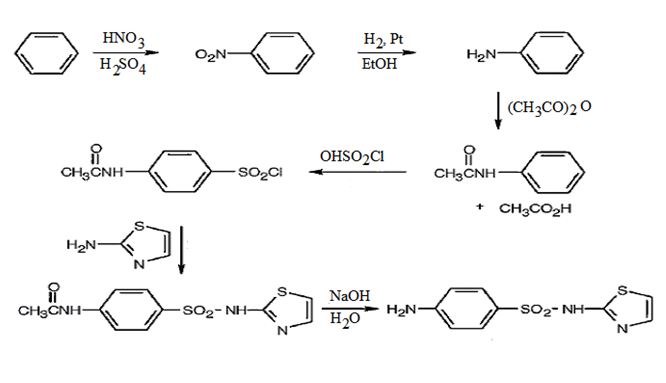
Explanation:
Nitration of benzene with Conc. HNO3 and Conc. H2SO4 yields nitrobenzene which on reduction with H2/Pt in ethanol gives aniline. Acetylation of aniline with acetic anhydride produces acetanilide. Chlorosulfonation of acetanilide leads to the formation of a p-chlorosulfonate as anilide group is orthp and para directing. Treatment of chlorosulfonate with 2-aminothiazole yields N-acetylsulfothiazole. Hydrolysis of N-acetylsulfothiazole yields sulfathiazole.
Conclusion:
The synthesis of sulfathiazole starting from benzene and using 2-aminothiazole is given below.

Trending nowThis is a popular solution!

Chapter 24 Solutions
Organic Chemistry
- Ibufenac, a para-disubstituted arene with the structure HO2CCH2C6H4CH2CH(CH3)2, is a much more potent analgesic than aspirin, but it was never sold commercially because it caused liver toxicity in some clinical trials. Devise a synthesis of ibufenac from benzene and organic halides having fewer than five carbons.arrow_forward3,5-Dibromo-4-hydroxybenzenesulfonic acid is used as a disinfectant. Propose a synthesis of this compound from phenol.arrow_forwardShow how to synthesize the following amines from the indicated starting materials byacylation–reduction. b) N-benzylaniline from anilinearrow_forward
- Propose a synthesis for propoxyphene from 1-phenyl-1-propanone and any other necessary reagents.arrow_forwardWhat carboxylic acid and amine are needed to synthesize the pain reliever phenacetin? Phenacetin was once a component of the over-the-counter pain reliever APC (aspirin, phenacetin, caffeine), but it is no longer used because of its kidney toxicity.arrow_forwardPropose the synthesis for the following transformations.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


