
Concept explainers
a)
Interpretation:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above, is to be shown.
Concept introduction:
Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To show:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above.
b)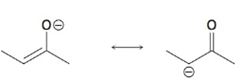
Interpretation:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above, is to be shown.
Concept introduction:
Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To show:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above.
c)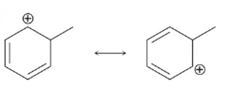
Interpretation:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above, is to be shown.
Concept introduction:
Resonance forms differ only in the placement of their π and nonbonding valence electrons. Neither the position nor the hybridization of any atom changes from one resonance form to another. The shift of electrons to give another resonance structure is represented by a curved arrow.
To show:
Using the curved-arrow formalism the flow of electrons in the resonance form on the left to give the one on the right, as given above.
Trending nowThis is a popular solution!

Chapter 2 Solutions
Organic Chemistry
- Define the consequence of resonance stabilization ?arrow_forwardCan someone please show me how the all the transfer of electrons occurs using arrows and writing out the formal charges. I'm particularly confused about the first line. Please show every single electron pair involved (even on the h2o).arrow_forwardWrite an equation for the proton transfer reaction that occurs when the following acid reacts with water. Draw curved arrows that show a mechanism for the proton transfer, and modify the given structures to draw the resulting products. Include lone pairs and formal charges.arrow_forward
- You will not find “hydroxide” in the stockroom, but you will find sodium hydroxide (NaOH) andpotassium hydroxide (KOH). Lithium hydroxide (LiOH) is expensive and used in spacecraft airfilters since hydroxide reacts with carbon dioxide, and lithium is lighter than sodium or potassium.Cesium and francium hydroxides are very expensive and little used. Is this information consistentwith your answer to the previous question?arrow_forwardWhat atom will have a formal charge in the following structure? CH O Atom A O Atom B Atom C O Atom Darrow_forwardDraw the conjugate base of C5F5H. Include lone pairs of electrons on all atoms. Add non-zero formal charges where appropriate.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

