
Concept explainers
Two molecules used in the
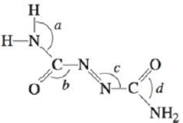
Azodicarbonamide
Methyl cyanoracy late
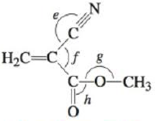
Azodicarbonamide is used in forming polystyrene. When added to the molten plastic, it decomposes to nitrogen, carbon monoxide, and ammonia gases, which are captured as bubbles in the molten polymer. Methyl cyanoacrylate is the main ingredient in super glue. As the glue sets, methyl cyanoacrylate
a. Complete the Lewis structures showing all lone pairs of electrons.
b. Which hybrid orbitals are used by the carbon atoms in each molecule and the nitrogen atom in azodicarbonamide?
c. How many π bonds are present in each molecule?
d. Give approximate values for the bond angles marked a through h in the previous structures.
Trending nowThis is a popular solution!

Chapter 4 Solutions
Chemistry: An Atoms First Approach
- Methylcyanoacrylate is the active ingredient in super glues. Its Lewis structure is (a) Give values for the three bond angles indicated. (b) Indicate the most polar bond in the molecule. (c) Circle the shortest carbon-oxygen bond. (d) Circle the shortest carbon-carbon bond.arrow_forwardwhat is the molecular geometry for the following compoundsarrow_forwardMark the correct statements related to the bonding, shape and polarity of molecules. A - Only valence electrons are used when Lewis structures are drawn. B - Valence electrons occupying pi bonding orbitals are not considered when determining the molecular shape with the VSEPR theory C- The lone pairs on the central atom does not influence the molecular shape of a molecule. D- Molecules with polar bonds are polar. E - Molecules with a tetrahedral electron pair geometry are non-polar.arrow_forward
- Ethylamine is an example of an important class of organiccompounds. The molecular formula of ethylamine isCH3CH2NH2. Draw its Lewis structure.arrow_forwardPlease count total valence electrons, have lewis structures, resonance structures when applicable, and name the molecular geometry for each central atom.arrow_forwardGroup X elements can conduct electricity and heat. As a chemistry student, Explain the characteristics of Group X elements. Explain the chemical properties of Group X elements . Discuss the trends of Group X elements when going down the group Using the knowledge of Lewis Diagram, show the formation of bond from any ONE of the element from Group X and any ONE non-metal element. State the electronic configuration of the cation and anion from the bond formed above. Draw the orbital diagram of the carbon and anion from thw bond formed above. Show the formation of positive ion and negative ion from the bond formed above and justify the differences in their number of electrons. Rubric: Part A; Choose your rubric based on the Bloom's. Part B; Assess originality.arrow_forward
- Dispersion forces are produced when an electronegative atom pulls on the electron of a hydrogen atom so much that the proton of the hydrogen can closely interact with the electronegative atom of another molecule electronegative atoms pull electrons toward one area of the molecule, creating regions of partial positive and partial negative charge that are attracted to regions of other molecules with the opposite charge electrons are transferred from one atom to another to produce anions and cations that are then held together in a rigid lattice electrons in molecular compounds are constantly moving and produce short-term regions of positive and negative charge that are attracted to regions of other molecules with the opposite chargearrow_forwardwhich of the following statement is not true? 1)Nitrogen s relative inertness is due to formation of strong and stable bond between 2 nitrogens atom. 2)the bond energy of N2 is relatively higher that makes it relatively inert. 3)the bond energy of an O-H bond is always the same in all compounds that contain an O-H bond 4)carbon-carbon single bond is the longest among all sorts of carbon-carbon bonds 5)a shorter bond in general ,is a stronger bond than a longer bondarrow_forwardDetermine the molecular structure of the following compound (C4H10O) using the data below Please show how you determined the structurearrow_forward
- Highlight the shorter bonds in the structure and describe their strength.arrow_forwardLewis Structure Electronic Geometry and Angles and Hybridization Identify the Polarity of the Molecule P, exception to Ocet Rule (Maximum of 10 electrons around P) PCI5 Electronic groups: Draw structure with the dipoles and Electronegativity values Count the electrons P is the central atom Octet Electronic Geometry: Valence Molecular Geometry: Shared Hybridization: Is it Symmetrical or Unsymmetrical? Bonds Bond Angles: Is it Polar or Non-polar Molecule? Lewis Structure Electronic Geometry and Identify the Polarity of the Molecule S, exception to Ocet Rule (Maximum of 12 electrons around S) Angles and Hybridization Electronic groups: Draw structure with the dipoles and Electronegativity values Count the electrons S is the central atom Octet Electronic Geometry: Valence Molecular Geometry: Shared Hybridization: Is it Symmetrical or Unsymmetrical? Bonds Bond Angles: Is it Polar or Non-polar Molecule?arrow_forwardWhy is water bent while Carbon dioxide is linear? Explain using the Lewis Representationarrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning





