
Introduction To General, Organic, And Biochemistry
12th Edition
ISBN: 9781337571357
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.2, Problem 4.2QC
Problem 4-2 Balance this equation:
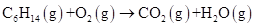
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
answer 5-6 please
balanced equation
type of chemical reaction
when the following equation is balance, the coefficient of H2 is K(s)+H2O(L)-->KOH(aq)+H2(g) answers options (a)1 (b)2 (c)3 (d)4 (e)5
Please answer 17-19 ( each given number to have a balanced equation and to identify its type of reaction)
Chapter 4 Solutions
Introduction To General, Organic, And Biochemistry
Ch. 4.2 - Problem 4-1 Following is an unbalanced equation...Ch. 4.2 - Problem 4-2 Balance this equation:Ch. 4.2 - Prob. 4.3QCCh. 4.3 - Problem 4-4 When a solution of copper(II)...Ch. 4.4 - Problem 4-5 In each equation, identify the...Ch. 4.5 - Problem 4-6 What is (a) the molecular weight of...Ch. 4.6 - Prob. 4.7QCCh. 4.6 - Problem 4-8 We wish to weigh 2.84 mol of sodium...Ch. 4.6 - Problem 4-9 How many moles of C atoms, H atoms,...Ch. 4.6 - Problem 4-10 How many moles of copper(I) ions,...
Ch. 4.6 - Prob. 4.11QCCh. 4.7 - Prob. 4.12QCCh. 4.7 - Prob. 4.13QCCh. 4.7 - Problem 4-14 Ethanol is produced industrially by...Ch. 4.7 - Prob. 4.15QCCh. 4.7 - Prob. 4.16QCCh. 4.8 - How many calories are required to heat 731g of...Ch. 4.8 - A 100g piece of iron at 25C is heated by adding...Ch. 4.8 - It required 88.2 cal to heat 13.4g of an unknown...Ch. 4.9 - Solid iron and oxygen gas react to form solid...Ch. 4 - 4-17 Balance each equation.Ch. 4 - 4-18 Balance each equation.Ch. 4 - Prob. 3PCh. 4 - 4-20 Calcium oxide is prepared by heating...Ch. 4 - 4-21 The brilliant white light in some firework...Ch. 4 - Prob. 6PCh. 4 - 4-23 When solid carbon burns in a limited supply...Ch. 4 - Prob. 8PCh. 4 - 4-25 In the chemical test for arsenic, the gas...Ch. 4 - Prob. 10PCh. 4 - Prob. 11PCh. 4 - 4-28 Answer true or false. (a) A net ionic...Ch. 4 - 4-29 Balance these net ionic equations. (a)...Ch. 4 - 4-30 In the equation (a) Identify the spectator...Ch. 4 - 4-31 Predict whether a precipitate will form when...Ch. 4 - 4-32 When a solution of ammonium chloride is added...Ch. 4 - 4-33 When a solution of hydrochloric acid, HCl, is...Ch. 4 - Prob. 18PCh. 4 - Prob. 19PCh. 4 - 4-36 Using the solubility generalizations given in...Ch. 4 - 4-37 Answer true or false. (a) When a substance is...Ch. 4 - Prob. 22PCh. 4 - Prob. 23PCh. 4 - Prob. 24PCh. 4 - Prob. 25PCh. 4 - 4-42 Calculate the formula weight of: (a) KCl (b)...Ch. 4 - 4-43 Calculate the molecular weight of: (a)...Ch. 4 - 4-44 Answer true or false. (a) The mole is a...Ch. 4 - 4-45 Calculate the number of moles in: (a) 32 g of...Ch. 4 - 4-46 Calculate the number of grams in: (a) 1.77...Ch. 4 - 4-47 Calculate the number of moles of: (a) O atoms...Ch. 4 - 4-48 Calculate the number of moles of: (a) S2-...Ch. 4 - 4-49 Calculate the number of: (a) nitrogen atoms...Ch. 4 - 4-50 How many molecules are in each of the...Ch. 4 - 4-51 What is the mass in grams of each number of...Ch. 4 - 4-52 The molecular weight of hemoglobin is about...Ch. 4 - 4-53 A typical deposit of cholesterol, C27H46O, in...Ch. 4 - 4-54 Answer true or false. (a) Stoichiometry is...Ch. 4 - 4-55 For the reaction: (a) How many moles of N2...Ch. 4 - 4-56 Magnesium reacts with sulfuric acid according...Ch. 4 - 4-57 Chloroform, CHCl3, is prepared industrially...Ch. 4 - 4-58 At one time, acetaldehyde was prepared...Ch. 4 - 4-59 Chlorine dioxide, ClO2, is used for bleaching...Ch. 4 - 4-60 Ethanol, C2H6O, is added to gasoline to...Ch. 4 - 4-61 In photosynthesis, green plants convert CO2...Ch. 4 - 4-62 Iron ore is converted to iron by heating it...Ch. 4 - Prob. 47PCh. 4 - 4-64 Aspirin is made by the reaction of salicylic...Ch. 4 - 4-65 Suppose the preparation of aspirin from...Ch. 4 - 4-66 Benzene reacts with bromine to produce...Ch. 4 - 4-67 Ethyl chloride is prepared by the reaction of...Ch. 4 - 4-68 Diethyl ether is made from ethanol according...Ch. 4 - Prob. 53PCh. 4 - How many calories are required to heat the...Ch. 4 - Prob. 55PCh. 4 - Prob. 56PCh. 4 - Prob. 57PCh. 4 - Prob. 58PCh. 4 - 4-71 Which of these reactions are exothermic, and...Ch. 4 - Prob. 60PCh. 4 - Prob. 61PCh. 4 - Prob. 62PCh. 4 - Prob. 63PCh. 4 - Prob. 64PCh. 4 - 4-77 To convert 1 mol of iron(III) oxide to its...Ch. 4 - 4-78 (Chemical Connections 4A) How does fluoride...Ch. 4 - Prob. 67PCh. 4 - Prob. 68PCh. 4 - 4-81 (Chemical Connections 4C) Balance the lithium...Ch. 4 - 4-82 When gaseous dinitrogen pentoxide, N2O5, is...Ch. 4 - Prob. 71PCh. 4 - Prob. 72PCh. 4 - Prob. 73PCh. 4 - 4-86 When an aqueous solution of Na3PO4 is added...Ch. 4 - Prob. 75PCh. 4 - 4-88 Chlorophyll, the compound responsible for the...Ch. 4 - 4-89 If 7.0 kg of is added to 11.0 kg of to form...Ch. 4 - 4-90 Lead(lI) nitrate and aluminum chloride react...Ch. 4 - 4-91 Assume that the average red blood cell has a...Ch. 4 - 4-92 Reaction of pentane, C5H12, with oxygen, O2,...Ch. 4 - 4-93 Ammonia is prepared industrially by the...Ch. 4 - 4-94 2,3,7,8-Tetrachlorodibenzo-p-dioxin (TCDD) is...Ch. 4 - Prob. 83PCh. 4 - Prob. 84PCh. 4 - Prob. 85PCh. 4 - Prob. 86PCh. 4 - Prob. 87PCh. 4 - Prob. 88PCh. 4 - Prob. 89PCh. 4 - Prob. 90PCh. 4 - Prob. 91PCh. 4 - 4-102 Aspartame, an artificial sweetener used as a...Ch. 4 - 4-103 Caffeine, a central nervous system...Ch. 4 - Prob. 94P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Problem 4-1 Following is an unbalanced equation for photosynthesis, the process by which green plants convert carbon dioxide and water to glucose and oxygen. Balance this equation:arrow_forwardProblem 7-53 Identify each of the following unbalanced reaction equations as belonging to the following categories: precipitation, acid-base, or oxidation-reduction. a. K2SO4(aq) + Ba(NO3)2(aq) → BaSO:(s) + KNO3(aq) b. HCl(aq) + Zn(s) H2(g) + ZnCl2(aq) c. HCl(aq) + AgNO3(aq) → HNO3(aq) + AgCl(s) d. HCl(aq) + KOH(aq) → H2O(1) + KCI(aq) e. Zn(s)+ CuSO«(aq)ZnSO4(aq) + Cu(s) f. NaH2PO4(aq) + NaOH(aq) Na3PO4(aq) + H2O(1) g. Ca(OH)2(aq) + H2SO4(aq) –→ CASO4(s) + H20(1) h. ZnCl2(aq) + Mg(s) → Zn(s) + MgCl2(aq)arrow_forwardQuestion 3 Balance the following reaction CO2 + H20 --> C6H1206 02arrow_forward
- r 5 Homework 1) Which chemical equation is properly balanced? A) SO2 + 02 + 4 H20→ 2 H2SO4 B) SO2 + 02 + H20→H2SO4 C) 4 SO2 + 02 + 4 H2O →4 H2S04 D) 2 SO2 + 02 + 2 H2O→2H2SO4 2) The chemical equation: KCIO3(s) KCI(s) select the statement that best describes the ba A) The balanced equation will have a coeffarrow_forwardb Lock Which of these does not represent a combination reaction? 82°F Flood Esc F1 ! 1 O Q metal oxide metal + oxygen A metal + oxygen metal oxide + water metal hydroxide F2 nonmetal + oxygen → nonmetal oxide 0: @ 2 W F3 S #M → 3 F4 E metal oxide D $ 4 F5 R % 5 F F6 ☆ T F7 ^ 6 G 다 Y F8 & 7 J+ H F9 U * 00 8 F10 L 12 Jarrow_forwardProblem 7-76 Write the balanced molecular, complete ionic, and net ionic equations for the reaction of ammonium sulfide with iron(III) chloride.arrow_forward
- Find the integer stoichiometry coefficients for the correctly balanced version of the following reaction: (Attachment) Options: (A) 1, 14, 1, 2, 1, 7 (B) 1, 14, 1, 1, 7 (C) 2, 1, 7, 1, 1, 5 (D) 1, 14, 9, 2, 7, 3arrow_forward1 OF 2 + 1 H₂O 8 OF₂ Reactants 3 H₂O 10₂ + 2 HF 0 0₂ 0 Products ? HF Check 0 OF₂ 0 Leftovers H₂Oarrow_forwardUse the References to access important values if needed for this question. When the following molecular equation is balanced using the smallest possible integer coefficients, the values of these coefficients are: iron (s) + hydrochloric acid (aq)- |iron(II) chloride (aq) + hydrogen (g) Submit Answer Try Another Version 2 item attempts remaining pt pt 1 pt Previous Nextarrow_forward
- Problem 7-22 For each of the following precipitation reactions, complete and balance the equation, indicating clearly which product is the precipitate. If no reaction would be expected (no precipitate), so indicate. a) NażCO3(aq) + K2SO4(aq) → b) CuCl2(aq) + (NH4)2CO3(aq) - c) K3PO4(aq)+ AIC\3(aq) –arrow_forwardGive the name of the missing organic reactant in the following reaction. ? + 2 Ag(NH3)2+ + 3OH¯ Submit Answer CH3~ Retry Entire Group 9 more group attempts remaining 0 + NH3 + 4 NH3 + 2 H₂O 2 Ag+ 4arrow_forwardQuestion 15 of 50 Submit Write a balanced chemical equation based on the following description: gaseous nitrogen and hydrogen react to form gaseous ammonia | 3C.2- Reset > 1 3 4 6. 7 8 9. + (s) (1) (g) (aq) H. Am NH4 Ni Ng Hg Ni Hy * H20 IIarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY