
Interpretation:
An efficient synthesis for the given transformation has to be predicted. The given reaction is shown as,
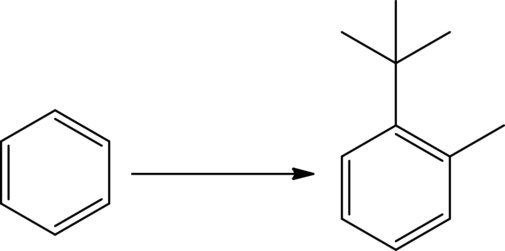
Concept Introduction:
Deactivators are electron withdrawing groups attached to the benzenes that have either positive charge or an atom with high electronegativity. They are meta directors.
Activators are electron donating groups attached to the benzenes that have either electron density that is able to push into benzene ring or a lone pair of electrons. They are ortho-para directing.
Halogens are deactivators that are ortho-para directing.
In a reaction, fuming sulfuric acid is used to introduce sulfonic groups in a compound (sulfonation) or sometimes used as blocking groups.
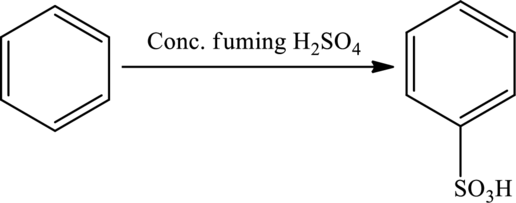
Chlorination: A reaction in which a chlorine atom is introduced to the compound.
In a reaction, sulfonic acid group can be removed from a compound by treating it with dilute sulfuric acid (desulfonation).
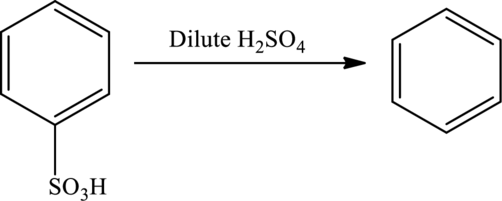
Friedel-Crafts Alkylation: This Lewis acid-catalyzed electrophilic
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
- Show all intermediates, mechanisms, and reagents. With each synthetic step in a true synthesis, you would need to analyze your product and confirm that you had isolated the correct intermediate or product. Often, a quick analysis is performed looking for a key signal that would confirm the desired product and confirm no starting material remains. For each step in your synthesis, explain how you would analyze the product to a) confirm product formation and b) confirm there is no remaining starting material. Your answer should clearly explain which analytical instrument you would use, what signal(s) you would look for and how that/those signal(s) would answer these questions. Br OH OH OHarrow_forwardSynthesize this compound. Your starting material should be phenol.arrow_forwardSeveral reagents and several organic structures are shown below. Construct a multistep synthetic route from the reactant 2-methyl-1-butene to the product 3-bromo-2-methyl-2-butanol by dragging the appropriate pieces into the bins. Note that each bin will hold only one item, and not every given reagent or structure will be used.arrow_forward
- Complete the reactions below using the required reagents or products. If there is no reaction, indicate so. If a product is in the majority, specify which one.arrow_forwardWhich reagent would be best for accomplishing the transformation shown below?arrow_forwardPropose a multi-step synthesis of the target molecule shown at the right, using the starting materials on the left and any other reagents you need. Show the reagents needed for each step and the product of each step. You will need 4 reaction arrows. Remember: Aldehyde + terminal alkyne anion in acid = alcoholarrow_forward
- The following problem is a synthesis challenge. You are provided with the starting material and final product. However, you are not provided with any reagents. You must determine all of that on your own. These CANNOT be accomplished in one step; it will take two or more steps to complete! You must show all necessary reagents and reaction products.arrow_forwardPropose an efficient synthesis for the following transformation. The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as “EBF”). If there is more than one correct solution, provide just one answer.arrow_forwardThis is another synthesis problem. Show reagents and intermediates synthesized along the way that allow you to produce the product from the given starting material. For this one do NOT assume you can isolate isomers - meaning - the product isomers shown must be the only predominant isomers you make during your synthesis! ? CH3OHarrow_forward
- 4. Provide a synthetic route to the following molecule using benzene and cyclohexane. Reagents cannot contain more than one carbon. Provide a mechanism for the last step of your synthetic route.arrow_forwardContinue the synthesis by selecting the reagents necessary for the next transformation shown. ? Br The transformation above can be performed with some reagent or combination of the reagents listed below. Give the necessary reagent(s) in the correct order, as a string of letters (without spaces or punctuation, such as "EBF"). If there is more than one correct solution, provide just one answer. A B C EtONa HBr, ROOR H₂, Lindlar's cat. D E F HBr HC=CNa H₂, Pd G H I 1) 03; 2) DMS 1) NaNH2; 2) EtBr 1) R₂BH; 2) H₂O2, NaOHarrow_forwardIn the reaction series below, write down the appropriate reagents that can be used where there are question marks.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
