
Concept explainers
Draw a Lewis structure and use VSEPR theory to determine the geometry of each molecule. If the molecule has more than one central atom, indicate the geometry about each of these and draw the three-dimensional structure.
Interpretation:
The Lewis structures for the given compounds are to be drawn and the geometry of each of the given molecules is to be determined and the geometry of more than one central atom is to be indicated using the VSEPR theory. The three-dimensional structures of the given molecules are to be drawn.
Concept Introduction:
According to the Lewis theory, in covalent bonds, atoms share their electrons. The steps for drawing a covalent Lewis structure are as follows:
Write the skeletal structure of the molecule.
Add the number of valence electrons of each of the atoms in the molecule to determine the total number of electrons in the molecule.
Place the electrons by dots to complete the octets of the atoms.
If the central atom has not obtained an octet, then its multiple bonds can be formed.
The VSEPR theory is helpful in predicting the shapes of molecules from their Lewis structures. The geometry of a molecule can be determined on the basis of the number of electron groups, lone pairs and bonding pairs in the molecule.
Answer to Problem 29E
Solution:
a)
Lewis structure:
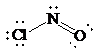
Molecular geometry is bent.
b)
Lewis structure:
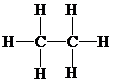
Molecular geometry is tetrahedral.
c)
Lewis structure:
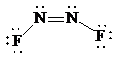
Molecular geometry is bent.
d)
Lewis structure:
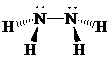
Molecular geometry is pyramidal.
Explanation of Solution
a)
The skeletal structure for the compound is as follows:
Now, the total number of electrons for the molecule is determined as follows:
Place the electrons as dots to give octet to each of the atoms in the molecule. Draw a single bond between the atoms. The central atom has not obtained an octet. Hence, nitrogen will form a double bond with oxygen to get its octet completed. Thus, the Lewis structure of
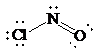
As all the atoms in the structure have obtained their octet, and thus, structure is complete. There are three electron pairs around the central atom. A double bond counts as a single electron group. Thus, there are two bonding pairs and one lone pair. Hence, according to the VSEPR theory, the molecular geometry is bent.
b)
The two carbon atoms are in the middle, each with three hydrogen atoms attached.
The skeletal structure for the compound is:
Now, the total number of electrons for the molecule is determined as follows:
Place the electrons as dots to give octet or duet to each of the atoms in the molecule. Draw a single bond between the atoms. Hence, the Lewis dot structure for the molecule will be as follows:
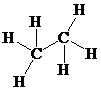
As all the hydrogen atoms in the structure have obtained their duet and both the carbon atoms have obtained their octet, thus, structure is complete. There are two central carbon atoms and so, the geometry is considered at each. There are four electron pairs around each carbon atom and no lone pair. Hence, according to the VSEPR theory, the molecular geometry is tetrahedral at each carbon atom.
c)
The two nitrogen atoms are in the centre and two fluorine atoms at the ends.
The skeletal structure for the compound is as follows:
Now, the total number of electrons for the molecule is determined as follows:
Place the electrons as dots to give octet to each of the atoms in the molecule. Draw a single bond between the atoms. Hence, the Lewis dot structure for the molecule will be as follows:
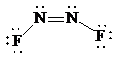
As all the atoms in the structure have obtained an octet, the structure is complete. There are two central nitrogen atoms and so, the geometry is considered at each. There are three electron groups around each nitrogen: two bonding groups and a lone pair. Hence, according to the VSEPR theory, the electron geometry is trigonal planar but the correct molecular geometry is bent.
d)
The two nitrogen atoms are in the centre and the two hydrogen atoms are attached to each nitrogen.
The skeletal structure for the compound is:
Now, the total number of electrons for the molecule is determined as follows:
Place the electrons as dots to give octet or duet to each of the atoms in the molecule. Draw a single bond between the atoms. Hence, the Lewis dot structure for the molecule will be as follows:
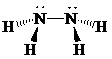
As all the hydrogen atoms in the structure have obtained their duet and the nitrogen atoms have completed their octets, the structure is complete. There are two central nitrogen atoms and so, the geometry is considered at each one. There are four electron groups around each nitrogen: three bonding groups and a lone pair. Hence, according to the VSEPR theory, the molecular geometry is trigonal pyramidal at each nitrogen atom.
Want to see more full solutions like this?
Chapter 5 Solutions
Chemistry In Focus
- Explain why the HOH molecule is bent, whereas the HBeH molecule is linear.arrow_forwardWhy is the geometric structure of a molecule important, especially for biological molecules?arrow_forwardA molecule has a Lewis structure where two atoms are connected to the central atom, and one lone pair is on the central atom. Each outer atom has three lone pairs attached to it. What is the shape of this molecule? a. Trigonal pyramid b. Trigonal planar c. Tetrahedral d. Bent e. Square planararrow_forward
- For each molecule below, draw the Lewis Dot diagram. Draw the 3D structure and determine the molecular shape of each. a. H2O b. NH3 c. CO2arrow_forwarda. Draw a Lewis structural diagram b. Using electronegativity, predict whether bonds will be polar or nonpolar c. Using bond polarity and shape, predict whether the molecule will be polar or nonpolar d. Identify the greatest Intermolecular force present Molecule CS₂ NH, Structural Diagram (include bond polarity and partial or full charges) Bond Type (ionic/polar covalent/ nonpolar covalent) Molecule Type (ionic/polar /nonpolar) Intermolecular Forcearrow_forwardA molecule is made from four atoms. The first atom (A) has four valence electrons, the second atom (B) has 6 valence electron, and the other two atoms (C) each have 7 valence electrons. What is the shape of the molecule ABC2? a. Tetrahedral d. Trigonal pyramid c. Trigonal planar d. Linear e. Bentarrow_forward
- 1. How many electrons will an iodine atom donate or accept, based on its number of valence electrons? A. Donate 7 electrons B. Donate 1 electron C. Accept 7 electrons D. Accept 1 electrons 2.What type of bond is formed between the two nitrogen atoms in diatomic nitrogen, N2? A. Triple Bond B. Double Covalent Bond C. Double Ionic Bond D. Single Bond 3.Which metal would form a stronger metallic bond? A. Lithium B. Sodium C. Strontium D. Tungsten 4. What holds the metal ions together in a lattice? A. Hydrogen Bonds B. Covalent Bonds C. Metallic Bonds D. Ionic Bondsarrow_forwardHave each member of your group select one of the molecules and complete steps a–d. Have members present their results to the rest of the group, explaining the reasoning they used to determine their answers. CS2 NCl3 CF4 CH2F2 a. Draw the Lewis structure.b. Determine the molecular geometry and draw it accurately.c. Indicate the polarity of any polar bonds within the structure.d. Classify the molecule as polar or nonpolar.arrow_forwardCHEMICAL BONDING A. Write the ionic-bond structure of the following compounds. Bal2 Ca3P2 SrSe B. Using the Pauling scale, determine the electronegativity difference to determine the kind of bond present in each compound. Compound Electronegativity Kind of Bond 1st Atom 2nd Atom Difference KI XeF2 BH3 ClO2 NiS C. Draw the Lewis electron dot structure and predict the shape of the following compounds/polyatomic ions. Molecule Total no. of e- Lewis structure Bonding Pairs Lone Pairs Geometry BrF3 PH3 PO3-3arrow_forward
- Beaulac Highline CHEM& 121 6) Double and triple bonds form because a. the atoms involved have high electronegativities. b. single covalent bonds do not give all of the atoms in the molecule 8 valence electrons. C. one of the atoms in the molecule has more than 8 valence electrons. d. the ions involved have charges larger than one. 0) Group IIA metals form ions with a charge. 8) Group VA nonmetals form ions with a charge. ) Group VIIA nonmetals form ions with a charge. ) List the metals not in group IA or IIA that only form one ion. ) Fill in the blanks in the following table: Polyatomic Ion Chemical Formula Polyatomic Ion Chemical Formula sulfate chlorate NO2 ammonium perbromate PO,3- CIO- nitrate bicarbonate cyanide iodite OH carbonatearrow_forwardWWW TTTTTT 23 How many unshared pairs of electrons are in one molecule? A. 1 pair B. 2 pairs Consider the covalent compound NF3. 24 How many total valence electrons are in one molecule? A. 22 C. 28 B. 26 D. 32 Consider the covalent compound NF3. 25 How many unshared pairs of electrons are in one molecule? A. 9 pairs B. 10 pairs C. 12 pairs D. 15 pairs 0: R HIC C H Study the four compound structures. One compound is correct and follows all of the rules for a stable covalent compound. Three compounds are incorrect and violate the 26 rules. Which structure is correct? A. B. HICIH H C. 3 pairs D. 4 pairs H-C C H H C. H 80%. H=C-C D. BLIVEWORKSHEETS H H H HO: H-C-C-H H Harrow_forward2. Avogadro does not "waste" his time drawing a Lewis structure before determining the shape of PF3. He thinks that the shape of PF3 must be trigonal planar because there are three fluorine atoms bonded to the central phosphorus atom. a. Draw the Lewis structure for PF3. b. Was Avogadro's answer for the shape of a PF3 molecule correct? Explain c. Why is it important to draw the Lewis structure for a molecule before identifying the shape of the molecule?arrow_forward

 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning



